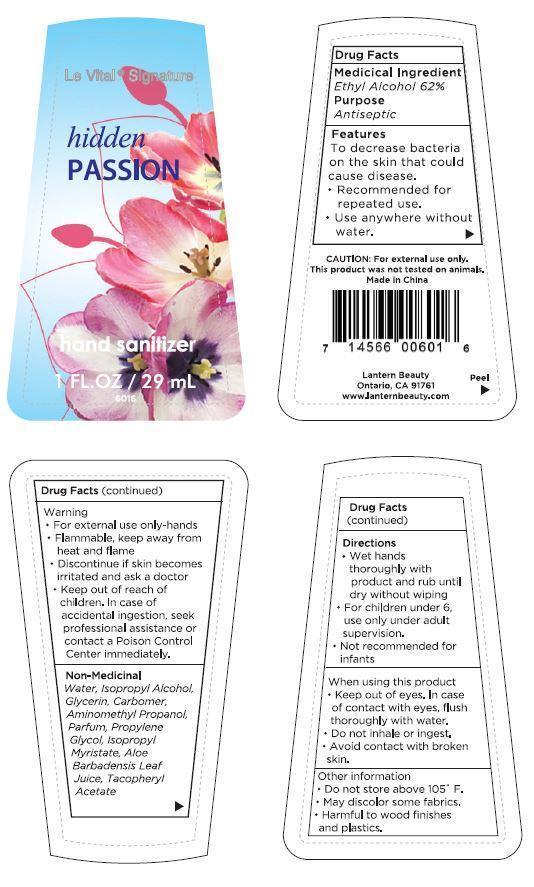
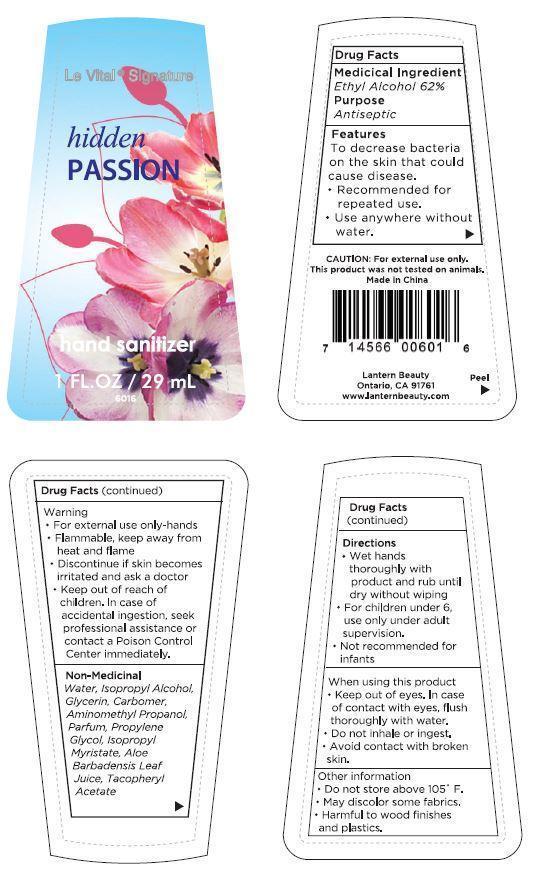
Label: LE VITAL HIDDEN PASSION- ethyl alcohol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 50306-001-01 - Packager: Shanghai Lantern Cosmetic Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 31, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
-
WARNINGS
Warning
- For external use only - hands
- Flammable, keep away from heat and flame
- Discontinue if skin becomes irritated and ask a doctor
CAUTION: For external use only. This product was not tested on animals.
When using this product
- Keep out of eyes. In case of contact with eyes, flush thoroughly with water.
- Do not inhale or ingest.
- Avoid contact with broken skin.
Other information
- Do not store above 105°F.
- May discolor some fabrics.
- Harmful to wood finishes and plastics.
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LE VITAL HIDDEN PASSION
ethyl alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50306-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ISOPROPYL ALCOHOL (UNII: ND2M416302) GLYCERIN (UNII: PDC6A3C0OX) CARBOMER COPOLYMER TYPE A (UNII: 71DD5V995L) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50306-001-01 29 g in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 07/23/2015 Labeler - Shanghai Lantern Cosmetic Co., Ltd. (421252043) Establishment Name Address ID/FEI Business Operations Shanghai Lantern Cosmetic Co., Ltd. 421252043 manufacture(50306-001)