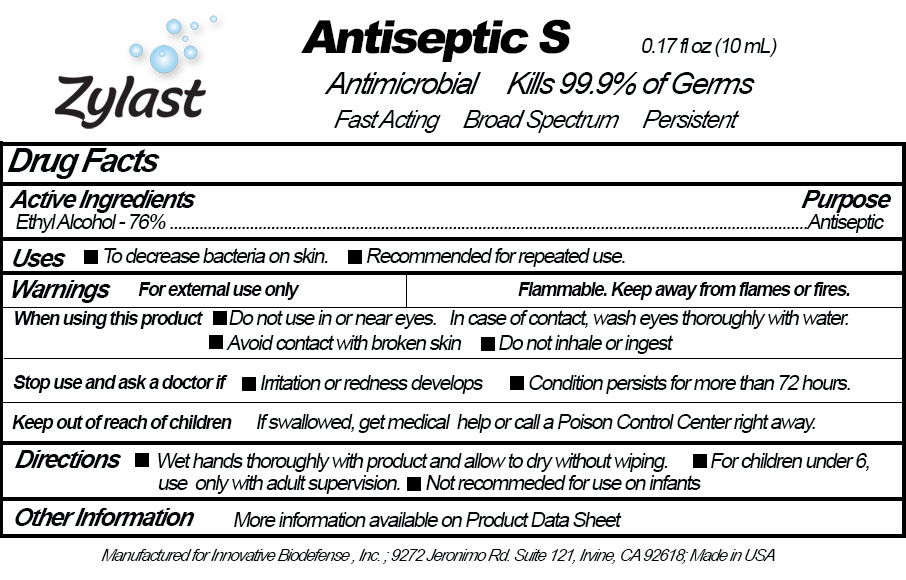
Label: ZYLAST ANTISEPTIC S- ethyl alcohol liquid
- NDC Code(s): 57362-466-16, 57362-466-17, 57362-466-41
- Packager: Innovative BioDefense
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- Uses
- Warning Section
- WHEN USING
- ASK DOCTOR
- KEEP OUT OF REACH OF CHILDREN
- Directions
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ZYLAST ANTISEPTIC S
ethyl alcohol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57362-466 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 760 mL in 1000 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57362-466-16 1000 mL in 1 CARTRIDGE; Type 0: Not a Combination Product 01/15/2019 2 NDC:57362-466-17 10 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 01/15/2019 3 NDC:57362-466-41 10 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product 01/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 01/15/2019 Labeler - Innovative BioDefense (078363903)