Label: FEXOFENADINE HYDROCHLORIDE tablet
-
NDC Code(s):
55111-782-01,
55111-782-30,
55111-782-78,
55111-782-79, view more55111-782-90, 55111-783-01, 55111-783-24, 55111-783-28, 55111-783-30, 55111-783-60, 55111-783-78, 55111-783-79, 55111-783-90, 55111-784-01, 55111-784-05, 55111-784-07, 55111-784-15, 55111-784-18, 55111-784-23, 55111-784-28, 55111-784-29, 55111-784-30, 55111-784-35, 55111-784-40, 55111-784-43, 55111-784-45, 55111-784-59, 55111-784-75, 55111-784-78, 55111-784-79, 55111-784-90
- Packager: Dr. Reddy's Laboratories Limited
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 22, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
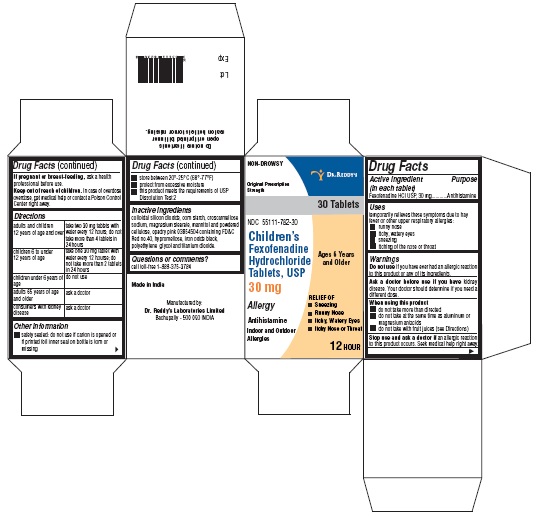
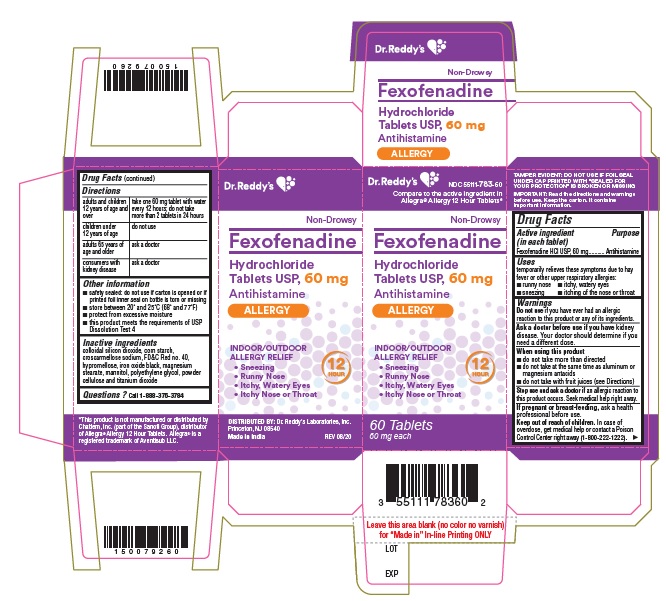
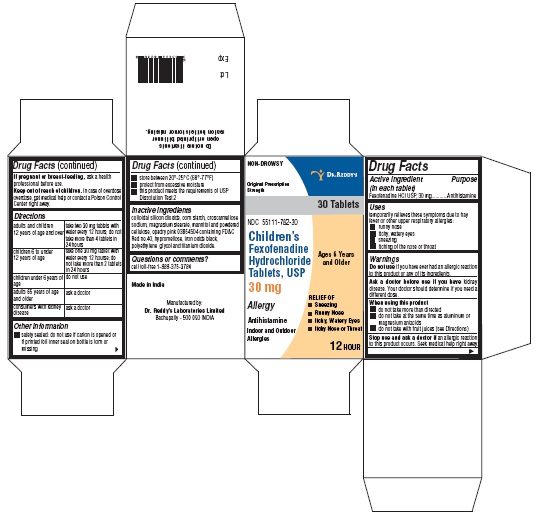
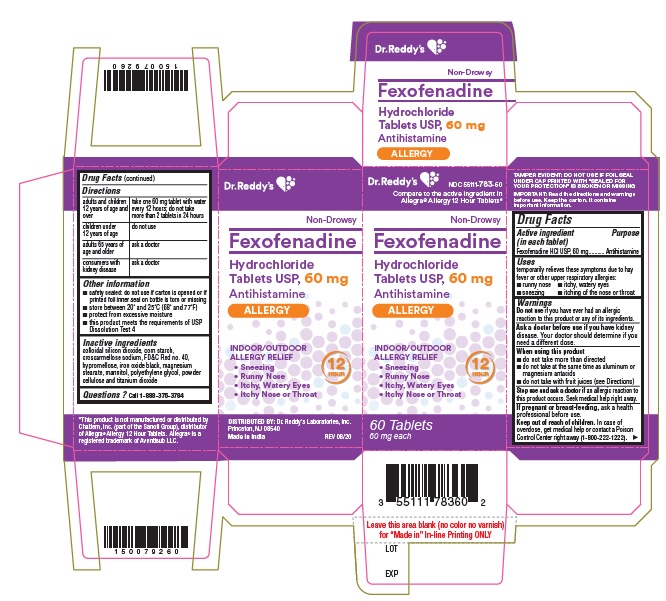
- Active ingredient(s)
- Purpose
- Use(s)
-
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
- Questions?
- PACKAGE LABEL PRINCIPAL DISPLAY PANEL SECTION
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55111-782 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Fexofenadine Hydrochloride (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) Fexofenadine Hydrochloride 30 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) POWDERED CELLULOSE (UNII: SMD1X3XO9M) FD&C RED NO. 40 (UNII: WZB9127XOA) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) FERROSOFERRIC OXIDE (UNII: XM0M87F357) polyethylene glycol 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color PINK Score no score Shape OVAL Size 4mm Flavor Imprint Code 192;R Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55111-782-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 2 NDC:55111-782-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 3 NDC:55111-782-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 4 NDC:55111-782-78 10 in 1 CARTON 01/03/2011 4 NDC:55111-782-79 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076502 01/03/2011 FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55111-783 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Fexofenadine Hydrochloride (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) Fexofenadine Hydrochloride 60 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) magnesium stearate (UNII: 70097M6I30) mannitol (UNII: 3OWL53L36A) POWDERED CELLULOSE (UNII: SMD1X3XO9M) FD&C RED NO. 40 (UNII: WZB9127XOA) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) FERROSOFERRIC OXIDE (UNII: XM0M87F357) polyethylene glycol 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color PINK Score no score Shape OVAL Size 5mm Flavor Imprint Code 193;R Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55111-783-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 2 NDC:55111-783-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 3 NDC:55111-783-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 4 NDC:55111-783-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 01/03/2011 5 NDC:55111-783-78 10 in 1 CARTON 01/03/2011 5 NDC:55111-783-79 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 6 NDC:55111-783-28 2 in 1 CARTON 12/01/2020 6 6 in 1 BLISTER PACK; Type 0: Not a Combination Product 7 NDC:55111-783-24 4 in 1 CARTON 12/01/2020 7 6 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076502 01/03/2011 FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55111-784 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Fexofenadine Hydrochloride (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) Fexofenadine Hydrochloride 180 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) POWDERED CELLULOSE (UNII: SMD1X3XO9M) FD&C RED NO. 40 (UNII: WZB9127XOA) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) FERROSOFERRIC OXIDE (UNII: XM0M87F357) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color PINK Score no score Shape OVAL Size 7mm Flavor Imprint Code 194;R Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55111-784-30 1 in 1 CARTON 01/03/2011 1 30 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:55111-784-43 2 in 1 CARTON 01/03/2011 2 30 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:55111-784-40 1 in 1 CARTON 01/03/2011 3 40 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:55111-784-45 1 in 1 CARTON 01/03/2011 4 45 in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC:55111-784-59 2 in 1 CARTON 01/03/2011 5 60 in 1 BOTTLE; Type 0: Not a Combination Product 6 NDC:55111-784-75 1 in 1 CARTON 01/03/2011 6 70 in 1 BOTTLE; Type 0: Not a Combination Product 7 NDC:55111-784-90 1 in 1 CARTON 01/03/2011 7 90 in 1 BOTTLE; Type 0: Not a Combination Product 8 NDC:55111-784-01 1 in 1 CARTON 01/03/2011 8 100 in 1 BOTTLE; Type 0: Not a Combination Product 9 NDC:55111-784-15 1 in 1 CARTON 01/03/2011 9 150 in 1 BOTTLE; Type 0: Not a Combination Product 10 NDC:55111-784-18 1 in 1 CARTON 01/03/2011 10 180 in 1 BOTTLE; Type 0: Not a Combination Product 11 NDC:55111-784-05 1 in 1 CARTON 01/03/2011 11 500 in 1 BOTTLE; Type 0: Not a Combination Product 12 NDC:55111-784-23 1 in 1 CARTON 01/03/2011 12 2 in 1 BLISTER PACK; Type 0: Not a Combination Product 13 NDC:55111-784-07 1 in 1 CARTON 01/03/2011 13 5 in 1 BLISTER PACK; Type 0: Not a Combination Product 14 NDC:55111-784-35 2 in 1 CARTON 12/01/2021 14 5 in 1 BLISTER PACK; Type 0: Not a Combination Product 15 NDC:55111-784-29 3 in 1 CARTON 01/03/2011 15 5 in 1 BLISTER PACK; Type 0: Not a Combination Product 16 NDC:55111-784-78 10 in 1 CARTON 01/03/2011 16 NDC:55111-784-79 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 17 NDC:55111-784-28 3 in 1 CARTON 01/03/2011 17 15 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076502 01/03/2011 Labeler - Dr. Reddy's Laboratories Limited (650562841) Establishment Name Address ID/FEI Business Operations Dr. Reddy's Laboratories Limited (FTO III) 918608162 analysis(55111-784, 55111-782, 55111-783) , manufacture(55111-784, 55111-782, 55111-783) Establishment Name Address ID/FEI Business Operations Reed-Lane, Inc. 001819879 pack(55111-782, 55111-783, 55111-784) Establishment Name Address ID/FEI Business Operations Quality Packaging Specialists International, LLC 080629831 pack(55111-784) Establishment Name Address ID/FEI Business Operations Dr. Reddy's Laboratories Louisiana, LLC 830397282 pack(55111-784) Establishment Name Address ID/FEI Business Operations DR. REDDY'S LABORATORIES LIMITED 860037244 analysis(55111-783) , manufacture(55111-783) , pack(55111-783, 55111-784)