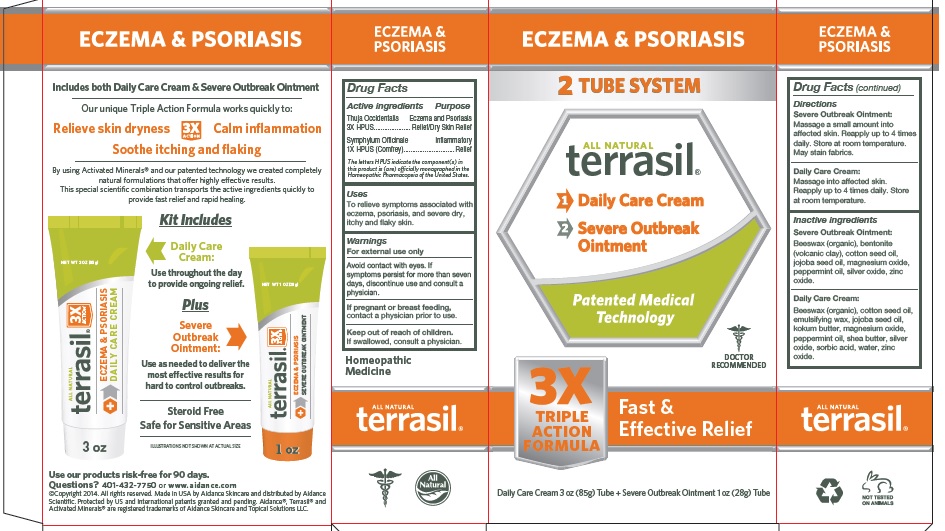
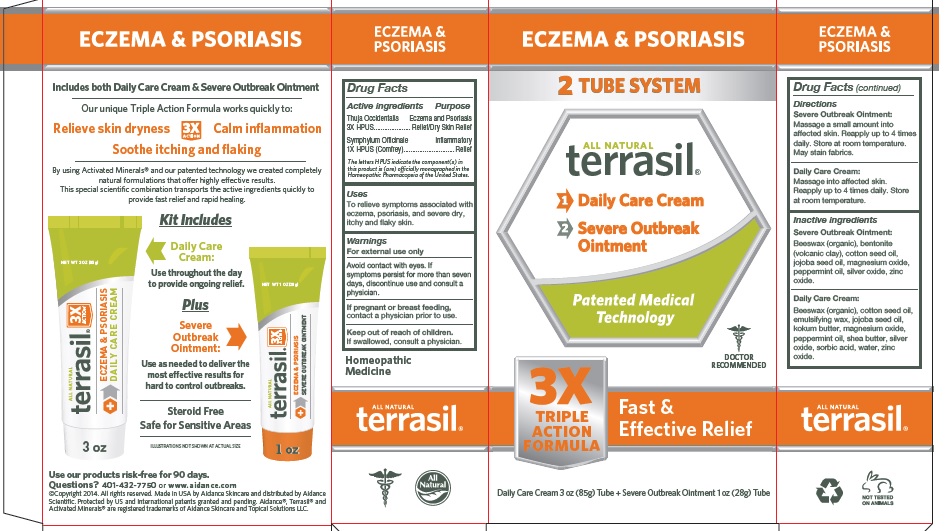
Label: TERRASIL ECZEMA AND PSORIASIS- thuja occidentalis, symphytum officinale kit
- NDC Code(s): 24909-167-99, 24909-177-85, 24909-187-28
- Packager: Aidance Skincare & Topical Solutions, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated April 11, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Directions
-
Inactive Ingredients
Severe Outbreak Ointment: Beeswax (organic), bentonite (volcanic clay), cotton seed oil, jojoba seed oil, magnesium oxide, peppermint oil, silver oxide, zinc oxide.
Daily Care Cream: Beeswax (organic), cotton seed oil, emulsifying wax, jojoba seed oil, kokum butter, magnesium oxide, peppermint oil, shea butter, silver oxide, sorbic acid, water, zinc oxide.
- PRINCIPAL DISPLAY PANEL - BOX
-
INGREDIENTS AND APPEARANCE
TERRASIL ECZEMA AND PSORIASIS
thuja occidentalis, symphytum officinale kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:24909-167 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24909-167-99 1 in 1 PACKAGE, COMBINATION 04/20/2016 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 85 g Part 2 1 TUBE 28 g Part 1 of 2 TERRASIL ECZEMA AND PSORIASIS DAILY CARE
thuja occidentalis, symphytum officinale creamProduct Information Item Code (Source) NDC:24909-177 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength THUJA OCCIDENTALIS LEAFY TWIG (UNII: 1NT28V9397) (THUJA OCCIDENTALIS LEAFY TWIG - UNII:1NT28V9397) THUJA OCCIDENTALIS LEAFY TWIG 3 [hp_X] in 1 g SYMPHYTUM OFFICINALE WHOLE (UNII: H8FJJ6KX5Y) (SYMPHYTUM OFFICINALE WHOLE - UNII:H8FJJ6KX5Y) SYMPHYTUM OFFICINALE WHOLE 1 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength WHITE WAX (UNII: 7G1J5DA97F) COTTONSEED OIL (UNII: H3E878020N) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) POLYSORBATE 60 (UNII: CAL22UVI4M) JOJOBA OIL (UNII: 724GKU717M) GARCINIA INDICA SEED BUTTER (UNII: US2H3D7800) MAGNESIUM OXIDE (UNII: 3A3U0GI71G) PEPPERMINT OIL (UNII: AV092KU4JH) SHEA BUTTER (UNII: K49155WL9Y) SILVER OXIDE (UNII: 897WUN6G6T) SORBIC ACID (UNII: X045WJ989B) ZINC OXIDE (UNII: SOI2LOH54Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24909-177-85 85 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/20/2016 Part 2 of 2 TERRASIL ECZEMA AND PSORIASIS SEVERE OUTBREAK
thuja occidentalis, symphytum officinale ointmentProduct Information Item Code (Source) NDC:24909-187 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength THUJA OCCIDENTALIS LEAFY TWIG (UNII: 1NT28V9397) (THUJA OCCIDENTALIS LEAFY TWIG - UNII:1NT28V9397) THUJA OCCIDENTALIS LEAFY TWIG 3 [hp_X] in 1 g SYMPHYTUM OFFICINALE WHOLE (UNII: H8FJJ6KX5Y) (SYMPHYTUM OFFICINALE WHOLE - UNII:H8FJJ6KX5Y) SYMPHYTUM OFFICINALE WHOLE 1 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength BENTONITE (UNII: A3N5ZCN45C) COTTONSEED OIL (UNII: H3E878020N) JOJOBA OIL (UNII: 724GKU717M) MAGNESIUM OXIDE (UNII: 3A3U0GI71G) PEPPERMINT OIL (UNII: AV092KU4JH) SILVER OXIDE (UNII: 897WUN6G6T) WHITE WAX (UNII: 7G1J5DA97F) ZINC OXIDE (UNII: SOI2LOH54Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24909-187-28 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/20/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/20/2016 Labeler - Aidance Skincare & Topical Solutions, LLC (018950611) Establishment Name Address ID/FEI Business Operations Aidance Skincare & Topical Solutions, LLC 018950611 manufacture(24909-167) , label(24909-167)