Label: ANTIMICROBIAL SKIN CLEANSER- chlorhexidine gluconate 2% solution liquid
- NDC Code(s): 21749-106-32, 21749-106-89
- Packager: Gojo Industries, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 18, 2019
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Uses
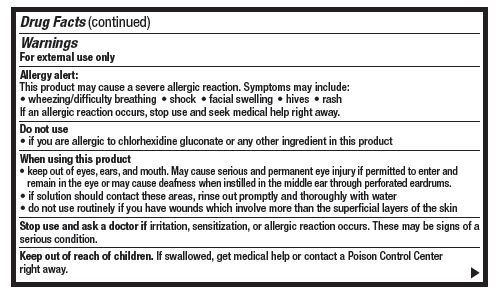
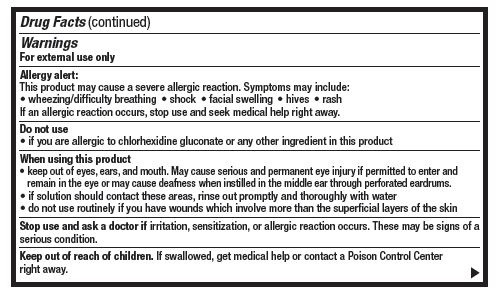
- Warnings
- Do not use
-
When using this product
- keep out of eyes, ears and mouth. May cause serious and permenant eye injury if permitted to enter and remain in the eye or may cause deafness when instilled through the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- do not use routinely if you have wounds which involve more than the superficial layers of the skin
- Stop use and ask a doctor if
- Keep out of reach of children
-
Directions
- use with care in premature infants under 2 months of age. These products may cause irritation or chemical burns.
Healthcare personnel handwash:
- wet hands with water
- dispense 5 mL of product (4 full strokes) into cupped hands and wash in a vigorous manner for 15 seconds
- dry thoroughly
Surgical hand scrub:
- wet hands and forearms with water
- scrub for 3 minutes with 5 mL of product (4 full strokes), with a wet brush, paying close attention to the nails, cuticles, and skin between the fingers
- a seperate nail cleaner may be used
- rinse thoroughly
- wash for an additional 3 minutes with 5 mL of product (4 full strokes) and rinse under running water and dry thoroughly
- OTHER SAFETY INFORMATION
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
NDC 21749-106-89
Chlorhexidine Gluconate 2% Solution
Antimicrobial SKin Cleanser
Distributed by:
Gojo Industries, Inc.
Akron, OH 44309
330-255-6000
www.gojo.com

NDC 21749-106-89
Chlorhexidine Gluconate 2% Solution
Antimicrobial SKin Cleanser
Distributed by:
Gojo Industries, Inc.
Akron, OH 44309
330-255-6000
www.gojo.com



NDC 21749-106-32
PROVON BRAND
Antimicrobial Skin Cleanser
Chlorhexidine Gluconate
2% Solution
Antiseptic
Distributed by:
GOJO Industries, Inc.
Akron, OH 44309
Questions?
Tel. 800-321-9647
*330-255-6000
www.GOJO.com
©2018. All rights reserved.
2PROV32BTLLBL
4104-12
Net Contents: 32 fl oz (1qt) (946 mL)



-
INGREDIENTS AND APPEARANCE
ANTIMICROBIAL SKIN CLEANSER
chlorhexidine gluconate 2% solution liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21749-106 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE GLUCONATE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) COCO DIETHANOLAMIDE (UNII: 92005F972D) HYDROXYETHYL CELLULOSE (2000 MPA.S AT 1%) (UNII: S38J6RZN16) ISOPROPYL ALCOHOL (UNII: ND2M416302) LAURAMINE OXIDE (UNII: 4F6FC4MI8W) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21749-106-89 1200 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2008 2 NDC:21749-106-32 946 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2008 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA019422 03/01/2008 Labeler - Gojo Industries, Inc. (004162038) Registrant - Xttrium Laboratories, Inc. (007470579) Establishment Name Address ID/FEI Business Operations Xttrium Laboratories, Inc. 007470579 manufacture(21749-106)