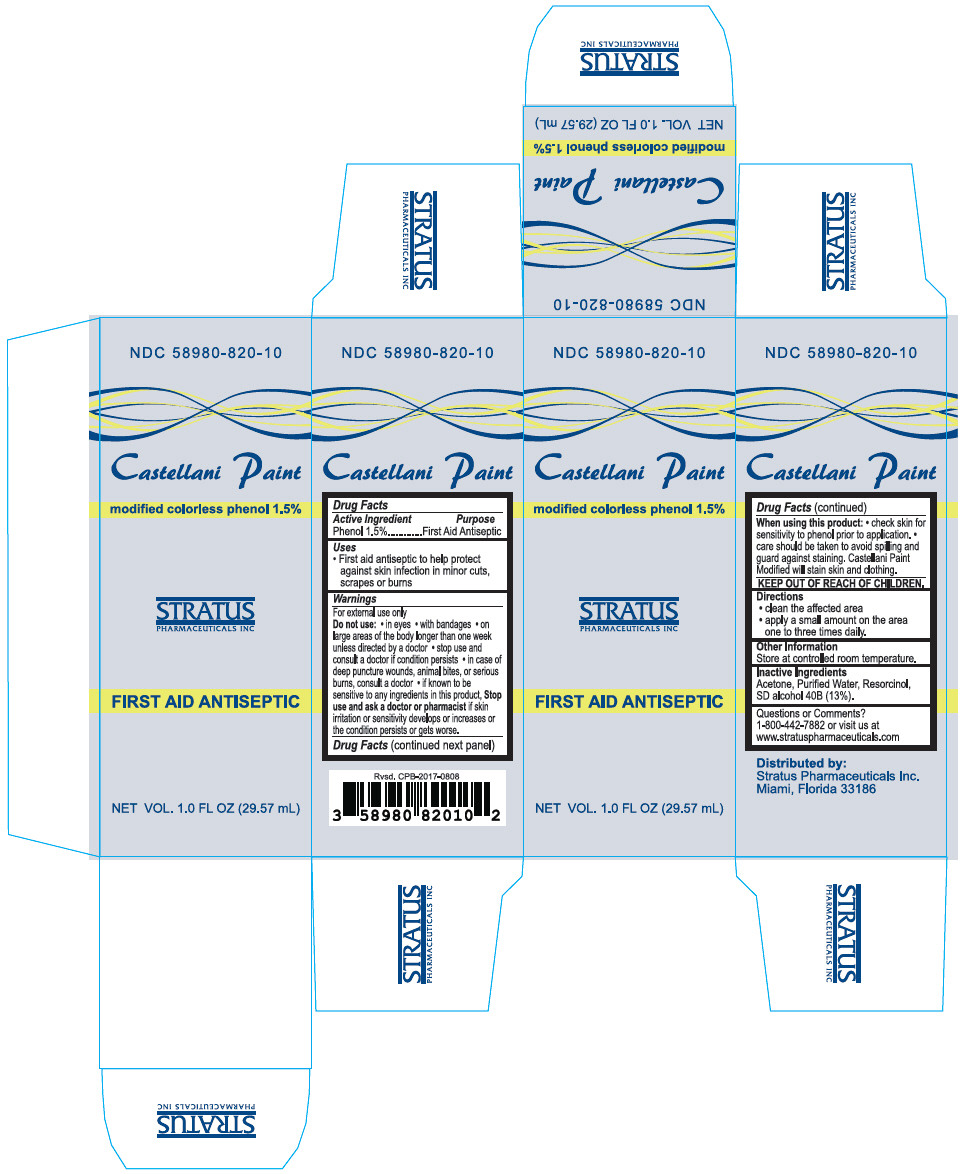
Label: CASTELLANI PAINT- phenol liquid
- NDC Code(s): 58980-820-10
- Packager: Stratus Pharamceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 19, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- in eyes
- with bandages
- on large areas of the body longer than one week unless directed by a doctor
- stop use and consult a doctor if condition persists
- in case of deep puncture wounds, animal bites, or serious burns, consult a doctor
- if known to be sensitive to any ingredients in this product,
Stop use and ask a doctor or pharmacist if skin irritation or sensitivity develops or increases or the condition persists or gets worse.
- Directions
- Other Information
- Inactive Ingredients
- Questions or Comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 29.57 mL Bottle Box
-
INGREDIENTS AND APPEARANCE
CASTELLANI PAINT
phenol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58980-820 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Phenol (UNII: 339NCG44TV) (Phenol - UNII:339NCG44TV) Phenol 15 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) ACETONE (UNII: 1364PS73AF) RESORCINOL (UNII: YUL4LO94HK) ALCOHOL (UNII: 3K9958V90M) Product Characteristics Color YELLOW (Clear to Yellow) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58980-820-10 1 in 1 BOX 04/01/2014 1 29.57 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part333A 04/01/2014 Labeler - Stratus Pharamceuticals (789001641) Establishment Name Address ID/FEI Business Operations TARMAC PRODUCTS INC 059890491 MANUFACTURE(58980-820) , LABEL(58980-820) , PACK(58980-820)