Label: TOPCARE MAXIMUM STRENGTH- benzocaine liquid
- NDC Code(s): 36800-443-58
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 7, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Allergy Alert: do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine or other "caine" anesthetics
When using this product
- avoid contact with eyes
- do not exceed recommended dosage
- do not use for more than 7 days unless directed by a dentist or doctor
-
DOSAGE & ADMINISTRATION
Directions
- Adults and children 2 years of age and older:
- apply to affected area using applicator tip
- use up to 4 times daily or as directed by a dentist or doctor
- children under 12 years of age should be supervised in the use of this product
- children under 2 years of age: consult a dentist or doctor
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
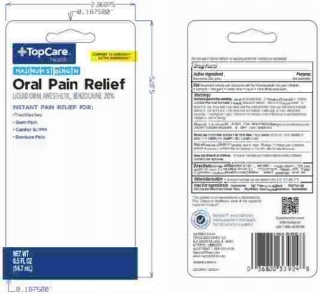
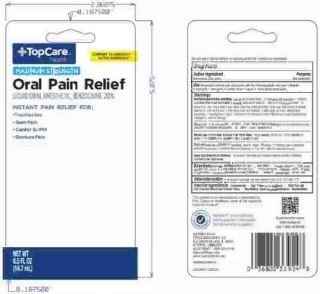
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOPCARE MAXIMUM STRENGTH
benzocaine liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-443 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 20 g in 100 g Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) SACCHARIN SODIUM (UNII: SB8ZUX40TY) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) METHYLPARABEN (UNII: A2I8C7HI9T) BENZYL ALCOHOL (UNII: LKG8494WBH) WATER (UNII: 059QF0KO0R) FD&C RED NO. 40 (UNII: WZB9127XOA) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Product Characteristics Color red (dark orange/red to sl brown) Score Shape Size Flavor MINT (N&A Mint Flavor 619179) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-443-58 1 in 1 BLISTER PACK 10/01/2014 1 14.17 g in 1 BOTTLE, WITH APPLICATOR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 10/01/2014 Labeler - Topco Associates LLC (006935977) Registrant - Lornamead Inc. (080046418) Establishment Name Address ID/FEI Business Operations HK KOLMAR CANADA, INC 243501959 manufacture(36800-443) , pack(36800-443)