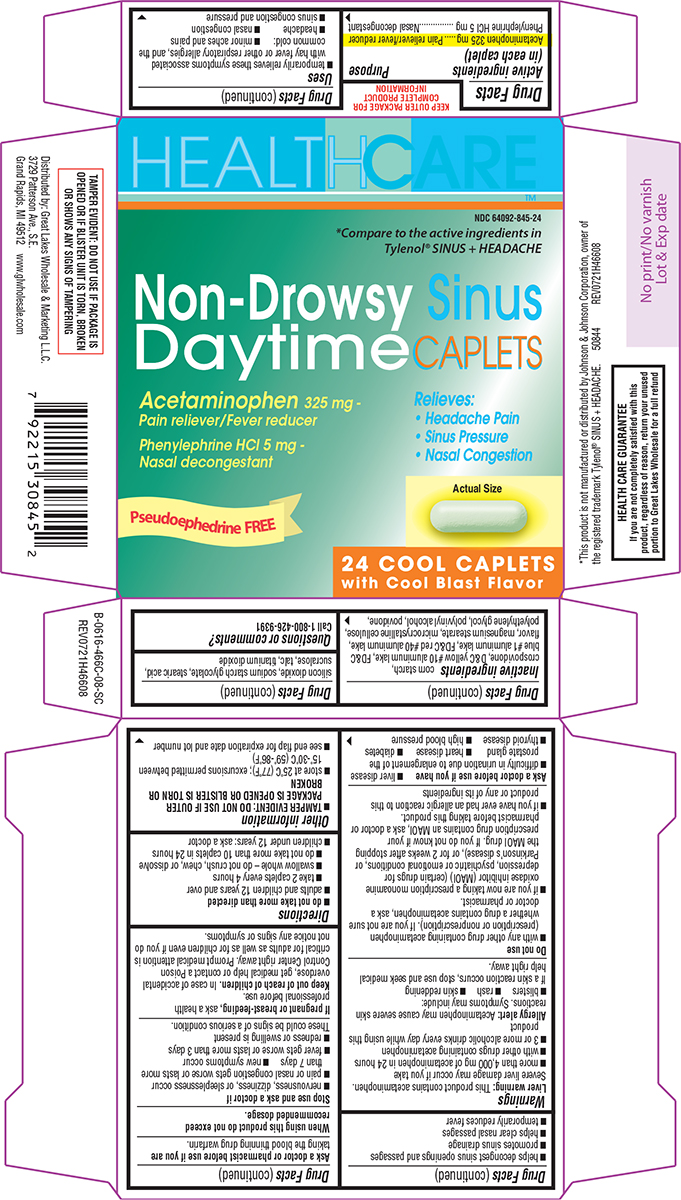
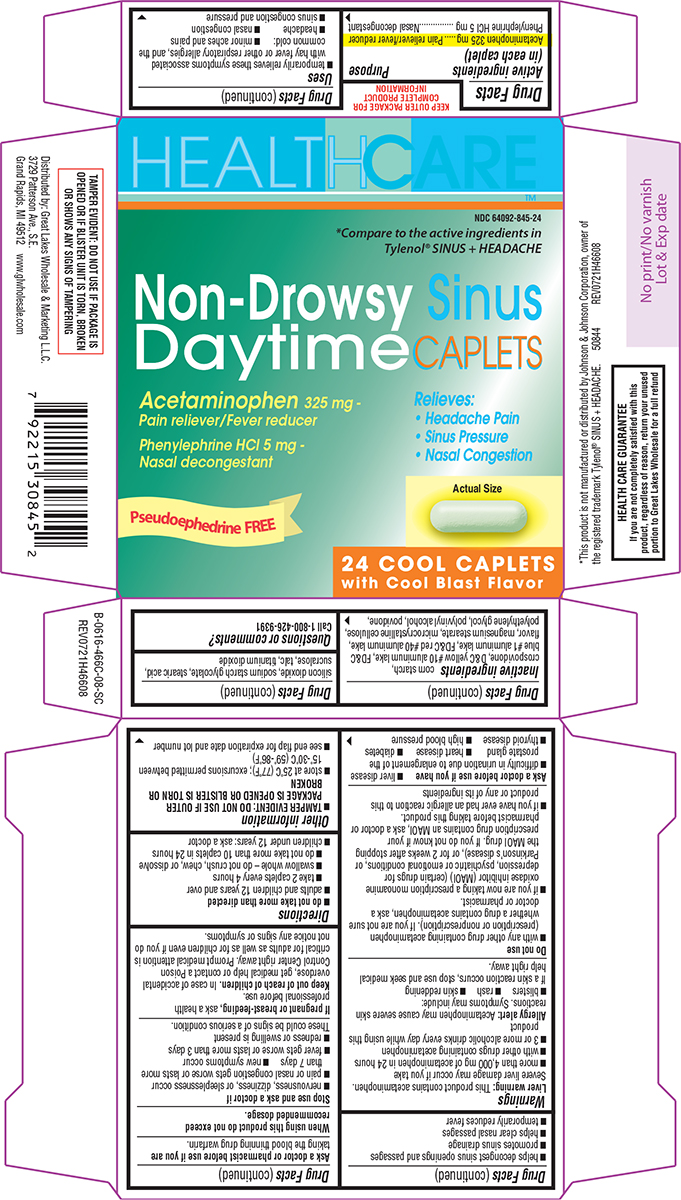
Label: NON-DROWSY SINUS DAYTIME- acetaminophen and phenylephrine hcl tablet, film coated
- NDC Code(s): 64092-845-24
- Packager: GREAT LAKES WHOLESALE, MARKETING, & SALES, INC.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 23, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each caplet)
- Purpose
-
Uses
- temporarily relieves these symptoms associated with hay fever or other respiratory allergies, and the common cold:
- minor aches and pains
- headache
- nasal congestion
- sinus congestion and pressure
- helps decongest sinus openings and passages
- promotes sinus drainage
- helps clear nasal passages
- temporarily reduces fever
- temporarily relieves these symptoms associated with hay fever or other respiratory allergies, and the common cold:
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- blisters
- rash
- skin reddening
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- liver disease
- difficulty in urination due to enlargement of the prostate gland
- heart disease
- diabetes
- thyroid disease
- high blood pressure
- Directions
- Other information
-
Inactive ingredients
corn starch, crospovidone, D&C yellow #10 aluminum lake, FD&C blue #1 aluminum lake, FD&C red #40 aluminum lake, flavor, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, sodium starch glycolate, stearic acid, sucralose, talc, titanium dioxide
- Questions or comments?
-
Principal Display Panel
HEALTHCARE™
NDC 64092-845-24
*Compare to the active ingredients in Tylenol®
SINUS + HEADACHE
Non-Drowsy Sinus
Daytime CAPLETSAcetaminophen 325 mg -
Pain reliever/Fever reducerPhenylephrine HCl 5 mg -
Nasal decongestantRelieves:
• Headache Pain
• Sinus Pressure
• Nasal CongestionPseudoephedrine FREE
24 COOL CAPLETS
with Cool Blast FlavorTAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING*This product is not manufactured or distributed by Johnson & Johnson Corporation, owner of
the registered trademark Tylenol® SINUS + HEADACHE. 50844 REV0721H46608Distributed by: Great Lakes Wholesale & Marketing L.L.C.
3729 Patterson Ave., S.E.
Grand Rapids, MI 49512 www.glwholesale.comHEALTH CARE GUARANTEE
If you are not completely satisfied with this
product, regardless of reason, return your unused
portion to Great Lakes Wholesale for a full refund
HealthCare 44-466C REV0721H
-
INGREDIENTS AND APPEARANCE
NON-DROWSY SINUS DAYTIME
acetaminophen and phenylephrine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64092-845 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) CROSPOVIDONE, UNSPECIFIED (UNII: 2S7830E561) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) FD&C RED NO. 40 (UNII: WZB9127XOA) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color green Score no score Shape OVAL Size 17mm Flavor MENTHOL Imprint Code 44;466 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64092-845-24 2 in 1 CARTON 07/26/2005 10/21/2024 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/26/2005 10/21/2024 Labeler - GREAT LAKES WHOLESALE, MARKETING, & SALES, INC. (361925498) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(64092-845) , pack(64092-845) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(64092-845) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(64092-845)