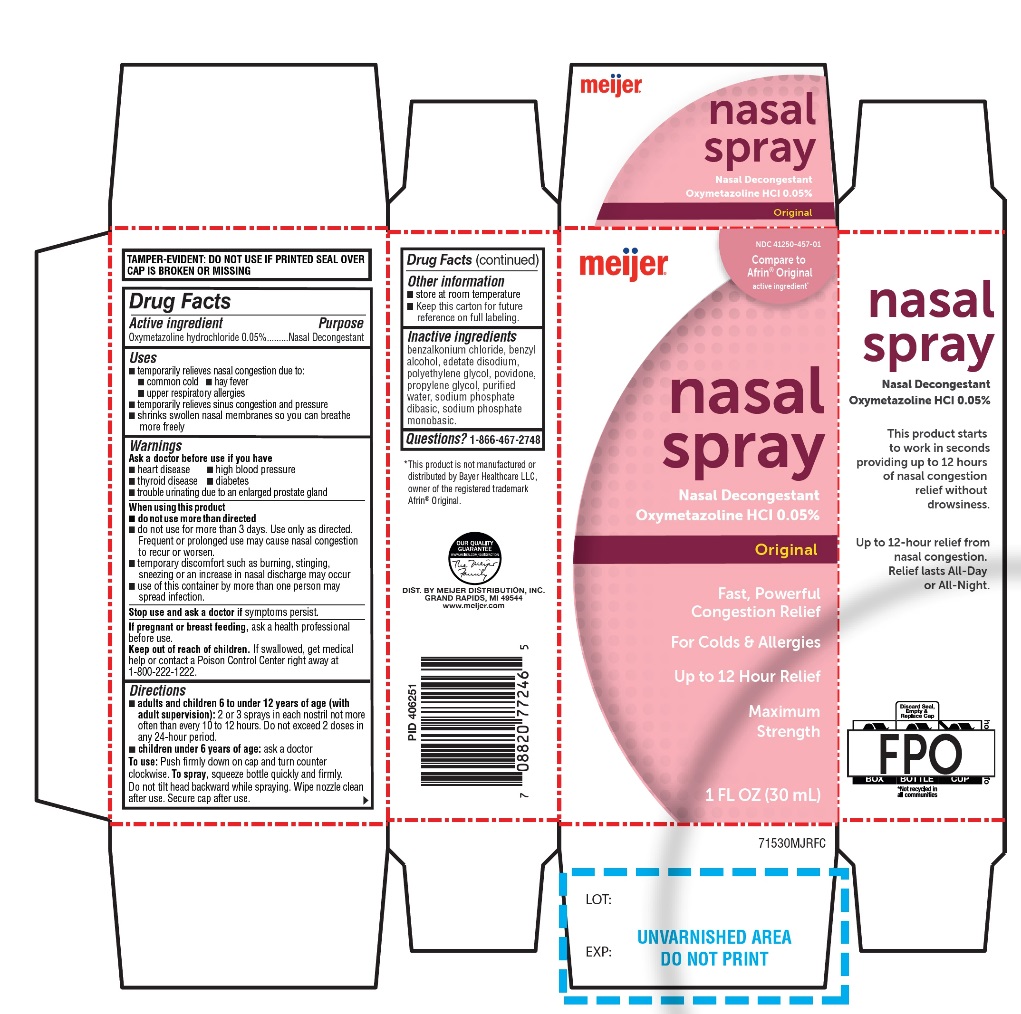
Label: MEIJER ORIGINAL- nasal spray
- NDC Code(s): 41250-457-01
- Packager: MEIJER DISTRIBUTION INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Ask a doctor before use if you have
-
When using this product
- ▪
- do not use more than directed
- ▪
- do not use for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen.
- ▪
- temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
- ▪
- use of this container by more than one person may spread infection
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
- ▪
- adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
- ▪
- children under 6 years of age: ask a doctor
To use: Push firmly down on cap and turn counter clockwise. To spray, squeeze bottle quickly and firmly. Do not tilt head backward while spraying. Wipe nozzle clean after use. Secure cap after use.
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
Compare to the active ingredient in Afrin® Original*
NDC# 41250-457-01
NASAL SPRAY
Nasal Decongestant
Oxymetazoline HCL 0.05%
ORIGINAL
Fast, Powerful Congestion Relief
For Colds & Allergies
Up to 12 Hour Relief
Maximum Strength
1 FL OZ (30 mL)
TAMPER-EVIDENT: DO NOT USE IF PRINTED SEAL OVER CAP IS BROKEN OR MISSING.
Distributed by:
*This product is not manufactured or distributed by Bayer Healthcare LLC, owner of the registered trademark Afrin® Original.
-
INGREDIENTS AND APPEARANCE
MEIJER ORIGINAL
nasal sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41250-457 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYMETAZOLINE HYDROCHLORIDE (UNII: K89MJ0S5VY) (OXYMETAZOLINE - UNII:8VLN5B44ZY) OXYMETAZOLINE HYDROCHLORIDE 0.05 g in 100 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BENZYL ALCOHOL (UNII: LKG8494WBH) EDETATE DISODIUM (UNII: 7FLD91C86K) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM PHOSPHATE, DIBASIC, UNSPECIFIED FORM (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, UNSPECIFIED FORM (UNII: 3980JIH2SW) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41250-457-01 1 in 1 CARTON 06/29/2020 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 06/29/2020 Labeler - MEIJER DISTRIBUTION INC (006959555)