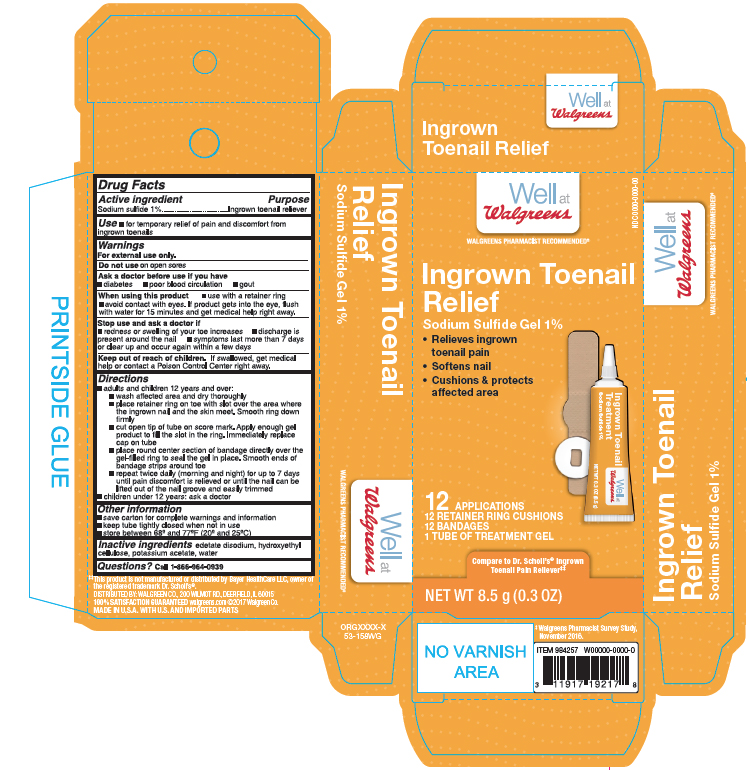
Label: SODIUM SUFIDE- ingrown toenail pain relief kit gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 0363-9560-85 - Packager: Walgreens Company
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 17, 2017
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Use
-
WARNINGS
For external use only.
When using this product
- use with a retainer ring
- avoid contact with eyes. If prodcut gets into the eye, flush with water for 15 minutes and get medical help reight away.
-
Directions
adults and children 12 yers and over:
- wash affected area and dry thoroghly
- place retainer ring on toe with slot over the area where the ingrown nail and the skin meet. Smooth ring down firmly
- cut open tip of the tube on score mark. Apply enough gel prodcut to fill the slot in the ring. Immeciately replace cap on tube
- place round center section of bandage directly over the gel- filled ring to seal the gel in place. Smooth ends of bandage strips around toe
- repeat twice daily (morning and night) for up to 7 days until pain discomfort is relieved or until the nail can be lifted out of the nail groove and easliy trimmed
Children under 12 years: ask a doctor
- Other Information
- Inactive Ingredient
- Questions?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SODIUM SUFIDE
ingrown toenail pain relief kit gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-9560 Route of Administration Topical Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM SULFIDE (UNII: YGR27ZW0Y7) (SULFIDE ION - UNII:G15I91XETI) SODIUM SULFIDE 0.085 g in 8.5 g Inactive Ingredients Ingredient Name Strength EDETATE DISODIUM (UNII: 7FLD91C86K) POTASSIUM ACETATE (UNII: M911911U02) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-9560-85 8.5 g in 1 PACKAGE; Type 0: Not a Combination Product 03/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358D 03/01/2018 Labeler - Walgreens Company (008965063)