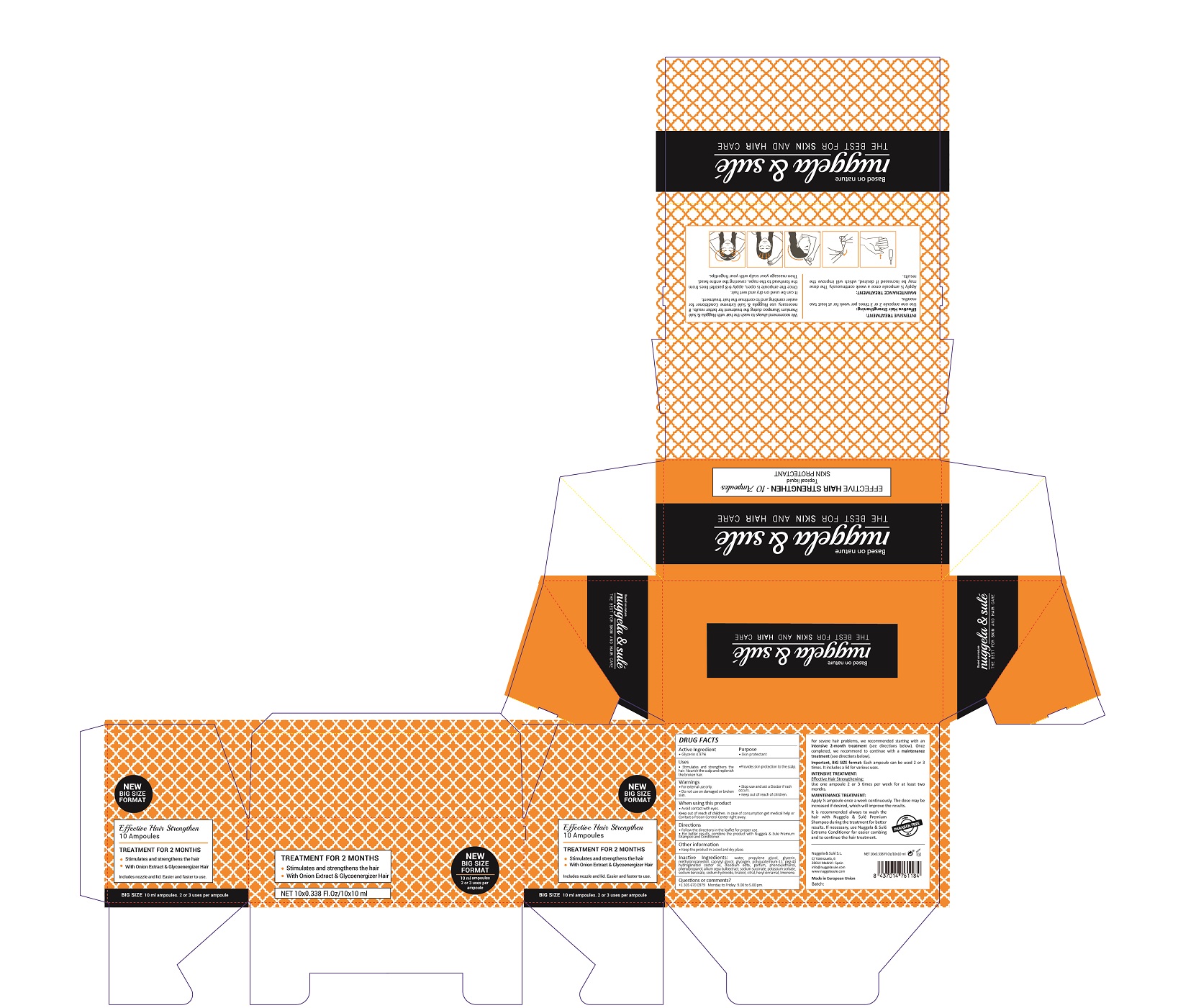
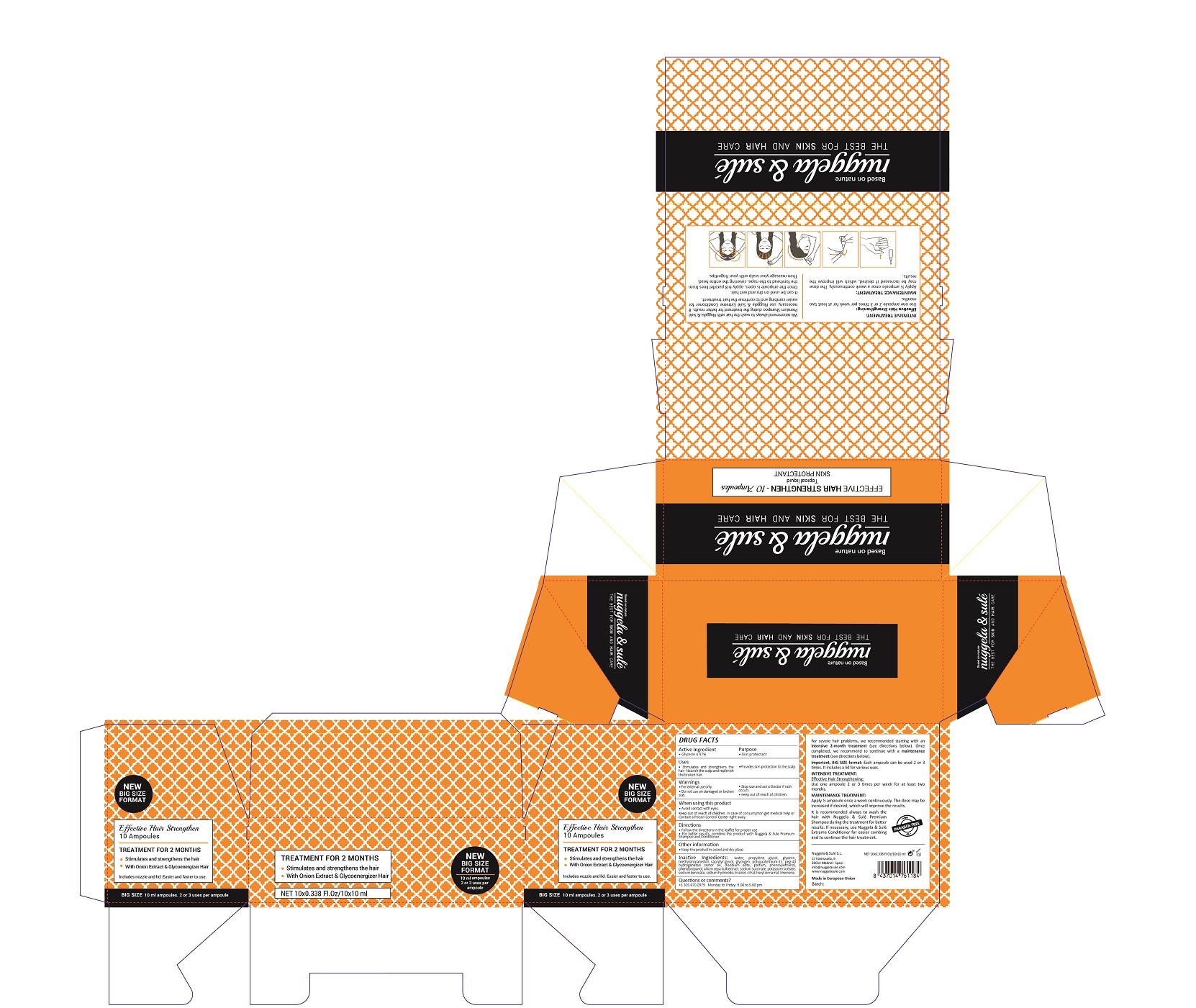
Label: EFFECTIVE HAIR STRENGTHEN- glycerin solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 71578-003-01, 71578-003-02 - Packager: NUGGELA & SULE SL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 28, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS PURPOSE
- Warnings
- Questions or comments?
- Other Information
- Directions
- Uses
-
Inactive Ingredients
AQUA, PROPYLENE GLYCOL, GLYCERIN, METHYLPROPANEDIOL, CAPRYLYL GLYCOL, GLYCOGEN, POLYQUATERNIUM-11, PEG-40 HYDROGENATED CASTOR OIL, DISODIUM EDTA , PARFUm, PHENOXYETHANOL, PHENYLPROPANOL, ALLIUM CEPA BULB EXTRACT, SODIUM SUCCINATE, POTASSIUM SORBATE, SODIUM BENZOATE, SODIUM HYDROXIDE, LINALOOL, CITRAL, HEXYL CINNAMAL, LIMONENE
- Uses
- Package Label
-
INGREDIENTS AND APPEARANCE
EFFECTIVE HAIR STRENGTHEN
glycerin solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71578-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 4.97 mg in 1 mL Inactive Ingredients Ingredient Name Strength LIMONENE, (+)- (UNII: GFD7C86Q1W) 0.0001 mg in 1 mL CITRAL (UNII: T7EU0O9VPP) 0.0001 mg in 1 mL LINALOOL, (-)- (UNII: 3U21E3V8I2) 0.0001 mg in 1 mL WATER (UNII: 059QF0KO0R) 84.31 mg in 1 mL CAPRYLYL GLYCOL (UNII: 00YIU5438U) 0.52 mg in 1 mL PHENOXYETHANOL (UNII: HIE492ZZ3T) 0.15 mg in 1 mL GLYCOGEN (UNII: 309GSC92U1) 0.5 mg in 1 mL POLYQUATERNIUM-11 (1000000 MW) (UNII: 0B44BS5IJS) 0.4 mg in 1 mL PEG-40 CASTOR OIL (UNII: 4ERD2076EF) 0.25 mg in 1 mL EDETATE DISODIUM (UNII: 7FLD91C86K) 0.2 mg in 1 mL PROPANEDIOL (UNII: 5965N8W85T) 6 mg in 1 mL PHENYLPROPANOL (UNII: 0F897O3O4M) 0.07 mg in 1 mL ONION (UNII: 492225Q21H) 0.04 mg in 1 mL POTASSIUM SORBATE (UNII: 1VPU26JZZ4) 0.005 mg in 1 mL SODIUM BENZOATE (UNII: OJ245FE5EU) 0.005 mg in 1 mL .ALPHA.-HEXYLCINNAMALDEHYDE (UNII: 7X6O37OK2I) 0.0001 mg in 1 mL SODIUM SUCCINATE HEXAHYDRATE (UNII: U16QOD6C4E) 0.01 mg in 1 mL SODIUM HYDROXIDE (UNII: 55X04QC32I) 0.004 mg in 1 mL METHYLPROPANEDIOL (UNII: N8F53B3R4R) 2.4 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71578-003-02 1 in 1 BOX 07/27/2017 1 NDC:71578-003-01 10 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 07/27/2017 Labeler - NUGGELA & SULE SL (466134663) Registrant - NUGGELA & SULE SL (466134663) Establishment Name Address ID/FEI Business Operations NUGGELA & SULE SL 466134663 manufacture(71578-003)