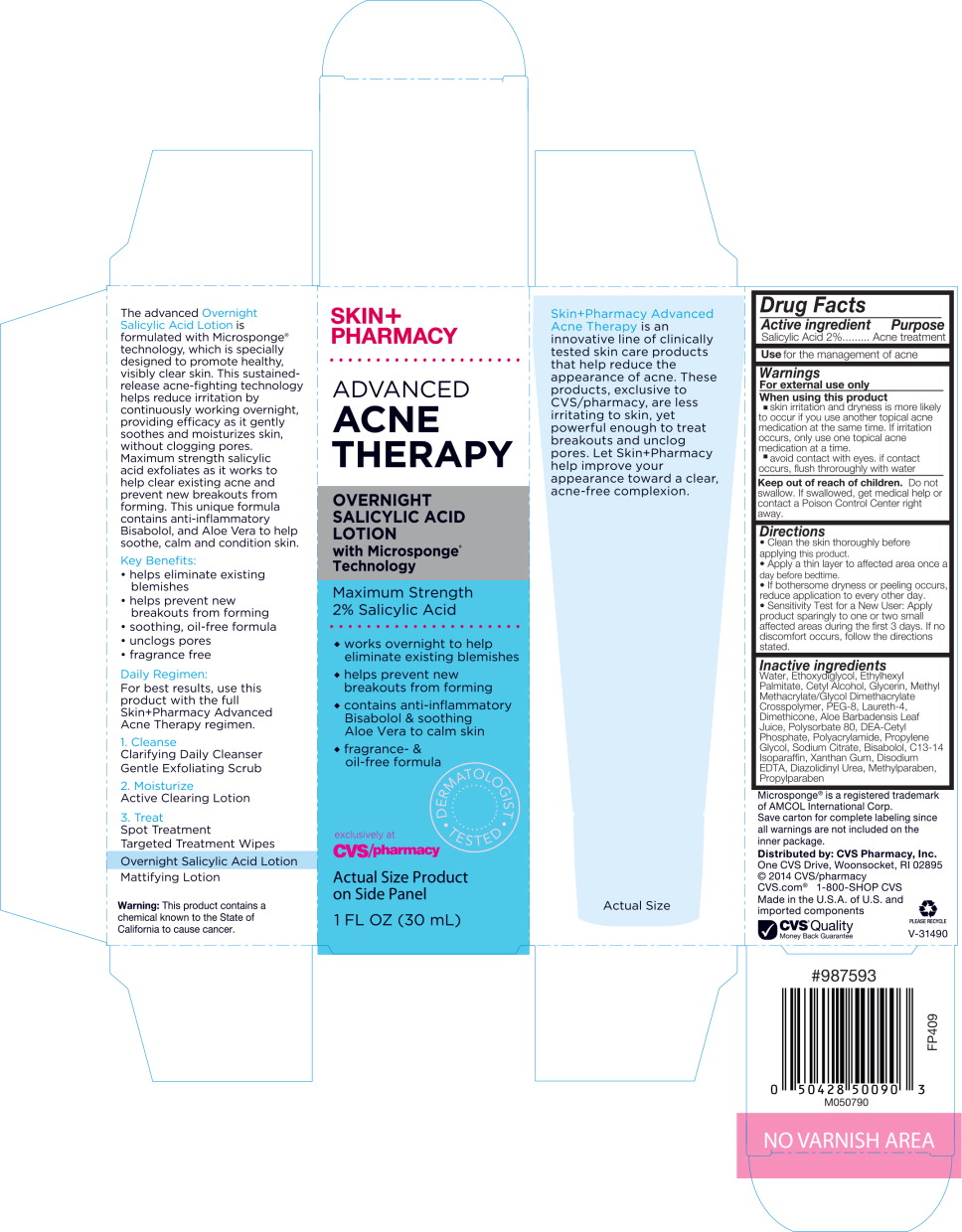
Label: SKIN PHARMACY ADVANCED ACNE THERAPY OVERNIGHT SALICYLIC ACID- salicylic acid liquid
- NDC Code(s): 68634-057-01
- Packager: AMCOL Health & Beauty Solutions, Inc. DBA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 11, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
HBS Code (Labeler): FP40901-30.2
Distributor: CVS Pharmacy, Inc.
Human OTC Drug
Active Ingredients: Salicylic Acid 2%
CARTON TEXT
________________________________________________________
SKIN+PHARMACY
ADVANCED ACNE THERAPY
OVERNIGHT SALICYLIC ACID LOTION with Microsponge® Technology
Maximum Strength 2% Salicylic Acid
- works overnight to help eliminate existing blemishes
- helps prevent new breakouts from forming
- contains anti-inflammatory Bisabolol & soothing Ale Vera to calm skin
- fragrance- & oil-free formula
DERMATOLOGIST TESTED
exclusively at CVS/pharmacy
Actual Size Product on Side Panel
1 FL OZ (30 mL)
The advanced Overnight Salicylic Acid Lotion is formulated with Microsponge® technology, which is specially designed to promote healthy, visibly clear skin. This sustained-release acne-fighting technology helps reduce irritation by continuously working overnight, providing efficacy as it gently soothes and moisturizes skin, without clogging pores. Maximum strength salicylic acid exfoliates as it works to help clear existing acne and prevent new breakouts from forming. This unique formula contains anti-inflammatory Bisabolol, and Aloe Vera to help soothe, calm and condition skin.
Key Benefits:
- helps eliminate existing blemishes
- helps prevent new breakouts from forming
- soothing, oil-free formula
- unclogs pores
- fragrance free
Daily Regiment:
For best results, use this product with the full Skin+Pharmacy Advanced Acne Therapy regimen.
- Cleanse
Clarifying Daily Cleanser Gentle Exfoliating Scrub - Moisturize
Active Clearing Lotion - Treat
Spot Treatment
Targeted Treatment Wipes
Overnight Salicylic Acid Lotion
Mattifying Lotion
Warning: This product contains a chemical known to the State of California to cause cancer.
Skin+Pharmacy Advanced Acne Therapy is an innovative line of clinically tested skin care products that help reduce the appearance of acne. These products, exclusive to CVS/pharmacy, are less irritating to skin, yet powerful enough to treat breakouts and unclog pores. Let Skin+Pharmacy help improve your appearance toward a clear, acne-free complexion.
Actual Size
Drug Facts
- Active Ingredient
- Purpose
- Use
-
Warnings
For external use only
-
Directions
- Clean the skin thoroughly before applying this product.
- Apply a thin layer to affected area once a day before bedtime.
- If bothersome dryness or peeling occurs, reduce application to every other day.
- Sensitivity Test for a New User: Apply product sparingly to one or two small affected areas during the first 3 days. If no discomfort occurs, follow the directions stated.
-
Inactive ingredients
Water, Ethoxydiglycol, Ethylhexyl Palmitate, Cetyl Alcohol, Glycerin, Methyl Methacrylate/Glycol Dimethacrylate Crosspolymer, PEG-8, Laureth-4, Dimethicone, Aloe Barbadensis Leaf Juice, Polysorbate 80, DEA-Cetyl Phosphate, Polyacrylamide, Propylene Glycol, Sodium Citrate, Bisabolol, C13-14 Isoparaffin, Xanthan Gum, Disodium EDTA, Diazolidinyl Urea, Methylparaben, Propylparaben
Microsponge® is a registered trademark of AMCOL International Corp.
Save carton for complete labeling since all warnings are not included on the inner package.
Distributed by: CVS Pharmacy, Inc.
One CVS Drive, Woonsocket, RI 02895
© 2014 CVS/pharmacy
CVS.com® 1-800-SHOP CVS
Made in the U.S.A. of U.S. and foreign components
V-13649CVS® Quality
Money Back GuaranteePLEASE RECYCLE
V-31490
#987593
FP409
M050790
________________________________________________________
end of text
160611
AMCOL Health & Beauty Solutions, Inc. DBA AMCOL Household and Personal Care
301 Laser Lane Lafayette, LA 70507 | www.amcolhpc.com
lauren.haase@mineralstech.com
-
Principal Display Panel - 20 g/2 mL Carton Label
SKIN +
PHARMACYADVANCED
ACNE
THERAPYOVERNIGHT
SALICYLIC ACID
LOTIONwith Microsponge*
TechnologyMaximum Strength
2% Salicylic Acid- works overnight to help
eliminate existing blemishes - helps prevent new
breakouts from forming - contains anti-inflammatory
Bisabolol & soothing
Aloe Vera to calm skin - fragrance- &
oil-free formula
exclusively at
CVS/pharmacy
Actual Size Product on Side Panel
1 FL OZ (30 mL)
- works overnight to help
-
Principal Display Panel - 20 g/2 mL Tube Label
SKIN +
PHARMACYADVANCED
ACNE
THERAPYOVERNIGHT
SALICYLIC ACID LOTIONwith Microsponge* Technology
Maximum Strength
2% Salicylic Acid- works overnight to help eliminate
existing blemishes - helps prevent new breakouts
from forming - contains anti-inflammatory
Bisabolol & soothing Aloe Vera
to calm skin - fragrance- & oil-free formula
exclusively at
CVS/pharmacy
1 FL OZ (30 mL)
- works overnight to help eliminate
-
INGREDIENTS AND APPEARANCE
SKIN PHARMACY ADVANCED ACNE THERAPY OVERNIGHT SALICYLIC ACID
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68634-057 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength salicylic acid (UNII: O414PZ4LPZ) (salicylic acid - UNII:O414PZ4LPZ) salicylic acid 20 g in 2 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIETHYLENE GLYCOL MONOETHYL ETHER (UNII: A1A1I8X02B) ETHYLHEXYL PALMITATE (UNII: 2865993309) CETYL ALCOHOL (UNII: 936JST6JCN) METHYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: EG97988M5Q) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) LAURETH-4 (UNII: 6HQ855798J) DIMETHICONE (UNII: 92RU3N3Y1O) ALOE VERA LEAF (UNII: ZY81Z83H0X) POLYSORBATE 80 (UNII: 6OZP39ZG8H) DIETHANOLAMINE CETYL PHOSPHATE (UNII: 4UG0316V9S) POLYACRYLAMIDE (1500 MW) (UNII: 5D6TC4BRWV) 1,2-PROPANEDIOL, 1-BENZOATE (UNII: K4K90ZQ89N) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) LEVOMENOL (UNII: 24WE03BX2T) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) XANTHAN GUM (UNII: TTV12P4NEE) EDETATE DISODIUM (UNII: 7FLD91C86K) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68634-057-01 1 in 1 CARTON 05/01/2014 1 30 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 05/01/2014 Labeler - AMCOL Health & Beauty Solutions, Inc. DBA (872684803) Establishment Name Address ID/FEI Business Operations AMCOL Health & Beauty Solutions, Inc. DBA 872684803 MANUFACTURE(68634-057) , PACK(68634-057) , LABEL(68634-057) , ANALYSIS(68634-057)