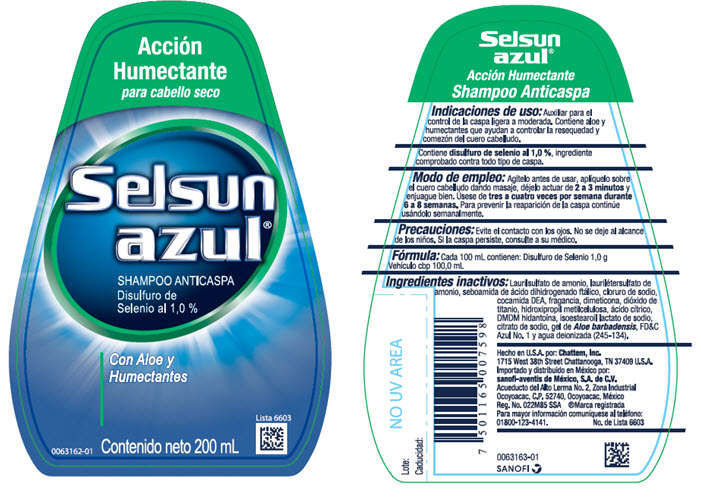
Label: SELSUN BLUE ACCION HUMETANTE- selenium sulfide shampoo
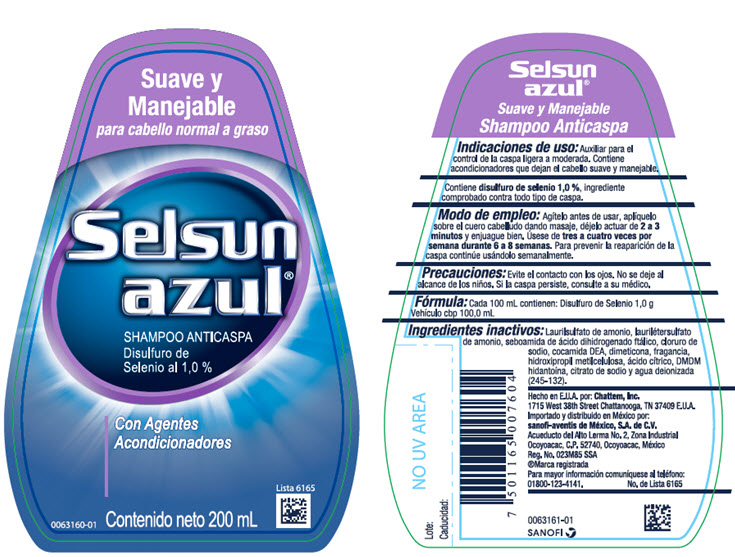
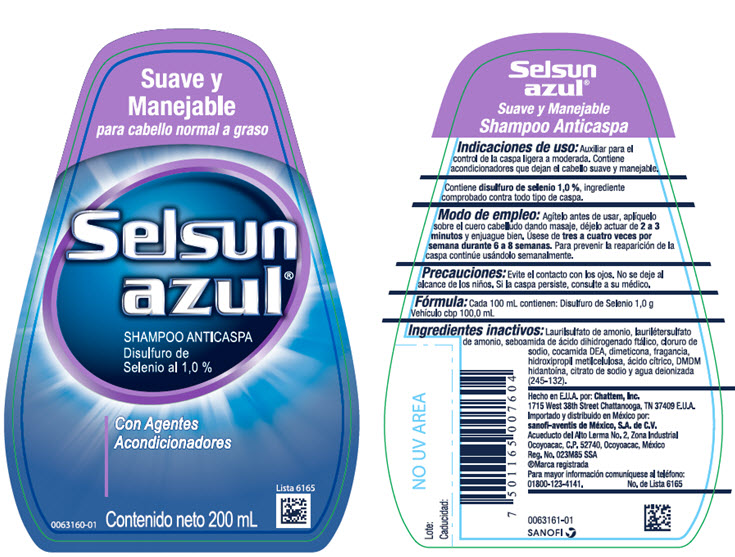
SELSUN AZUL SUAVE Y MANEJABLE HUMECTANTE- selenium sulfide shampoo
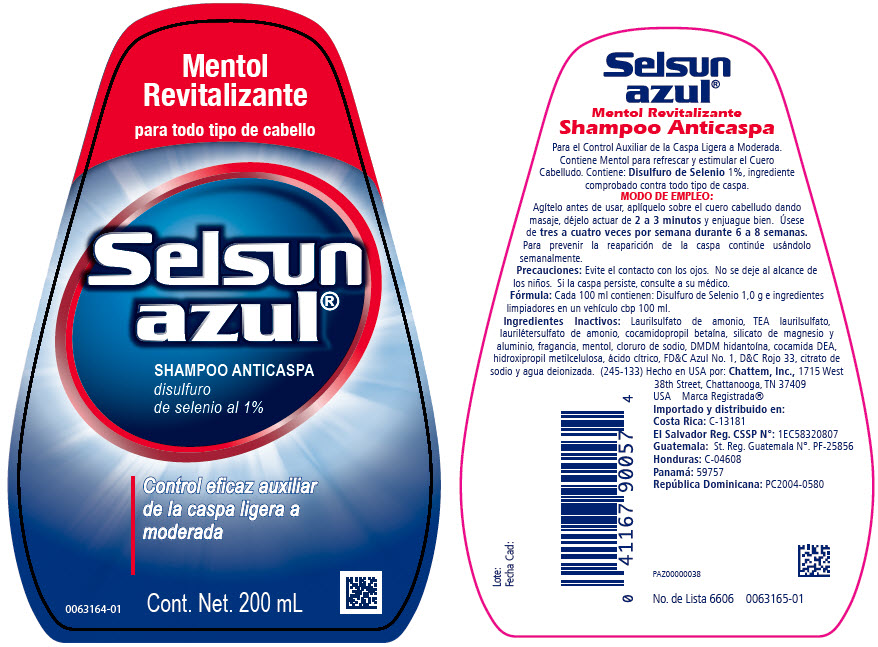
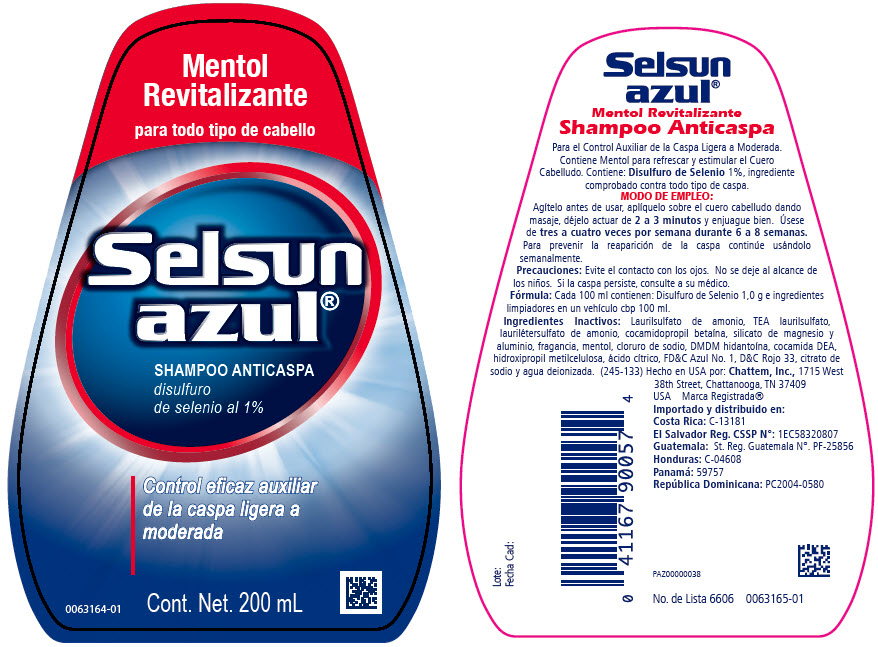
SELSUN AZUL REVITALIZANTE- selenium sulfide shampoo
-
Contains inactivated NDC Code(s)
NDC Code(s): 41167-0075-9, 41167-0076-0, 41167-9005-7 - Packager: Chattem, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated October 16, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Indicaciones de uso:
- Modo de empleo:
- Precauciones:
- Fórmula:
-
Ingredientes inactivos:
Laurilsulfato de amonio, laurilétersulfato de amonio, seboamida de ácido dihidrogenado ftálico, cloruro de sodio, cocamida DEA, fragancia, dimeticoa, dioxide de titanio, hidroxipropil metilcelulosa, ácido cítrico, DMDM hidantoína, isoestearoil lactate de sodio, citrato de sodio, gel de Aloe barbadensis, FD&C Azul No. 1 y agua deionizada (245-134).
- SPL UNCLASSIFIED SECTION
- Indicaciones de uso:
- Modo de empleo:
- Precauciones:
- Fórmula:
- Ingredientes inactivos:
- SPL UNCLASSIFIED SECTION
- PURPOSE
- Modo de empleo:
- Precauciones:
- Fórmula:
-
Ingredientes inactivos:
Laurilsulfato de amonio, TEA laurilsulfato, laurilétersulfato de amonio, cocamidopropil betaína, silicato de magnesio y aluminio, fragancia, mentol, cloruro de sodio, DMDM hidantoína, cocamida DEA, hidroxipropil metilcelulosa, ácido cítrico, FD&C Azul No. 1, D&C Rojo 33, citrato de sodio y agua deionizada.
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SELSUN BLUE ACCION HUMETANTE
selenium sulfide shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-0075 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SELENIUM SULFIDE (UNII: Z69D9E381Q) (SELENIUM SULFIDE - UNII:Z69D9E381Q) SELENIUM SULFIDE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) AMMONIUM LAURYL SULFATE (UNII: Q7AO2R1M0B) SULFATE ION (UNII: 7IS9N8KPMG) COCO DIETHANOLAMIDE (UNII: 92005F972D) DMDM HYDANTOIN (UNII: BYR0546TOW) HYPROMELLOSES (UNII: 3NXW29V3WO) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM CHLORIDE (UNII: 451W47IQ8X) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ALOE VERA LEAF (UNII: ZY81Z83H0X) DIMETHICONE (UNII: 92RU3N3Y1O) SODIUM ISOSTEAROYL LACTYLATE (UNII: 8730J0D3EV) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-0075-9 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date EXPORT ONLY 12/01/2012 SELSUN AZUL SUAVE Y MANEJABLE HUMECTANTE
selenium sulfide shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-0076 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SELENIUM SULFIDE (UNII: Z69D9E381Q) (SELENIUM SULFIDE - UNII:Z69D9E381Q) SELENIUM SULFIDE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) AMMONIUM LAURYL SULFATE (UNII: Q7AO2R1M0B) SULFATE ION (UNII: 7IS9N8KPMG) COCO DIETHANOLAMIDE (UNII: 92005F972D) DMDM HYDANTOIN (UNII: BYR0546TOW) HYPROMELLOSES (UNII: 3NXW29V3WO) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM CHLORIDE (UNII: 451W47IQ8X) DIMETHICONE (UNII: 92RU3N3Y1O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-0076-0 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date EXPORT ONLY 12/01/2012 SELSUN AZUL REVITALIZANTE
selenium sulfide shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-9005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SELENIUM SULFIDE (UNII: Z69D9E381Q) (SELENIUM SULFIDE - UNII:Z69D9E381Q) SELENIUM SULFIDE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) AMMONIUM LAURYL SULFATE (UNII: Q7AO2R1M0B) TRIETHANOLAMINE LAURYL SULFATE (UNII: E8458C1KAA) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) MENTHOL (UNII: L7T10EIP3A) COCO DIETHANOLAMIDE (UNII: 92005F972D) DMDM HYDANTOIN (UNII: BYR0546TOW) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) HYPROMELLOSES (UNII: 3NXW29V3WO) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM CHLORIDE (UNII: 451W47IQ8X) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-9005-7 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date EXPORT ONLY 12/01/2012 Labeler - Chattem, Inc. (003336013)