Label: NAUS-EASE- meclizine hydrochloride film, soluble
- NDC Code(s): 49467-104-01, 49467-104-16
- Packager: Sunascen Therapeutics LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
-
DESCRIPTION
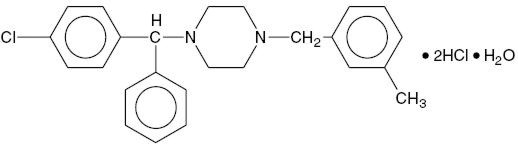
Chemically, Meclizine Hydrochloride, USP is 1-( p-Chloro-α-phenylbenzyl)-4-( m-methylbenzyl) piperazine dihydrochloride monohydrate. Meclizine Hydrochloride, USP is an oral antiemetic, which is a white to slightly yellowish crystalline powder having a slight odor and is tasteless. The molecular weight is 481.89 g/mol. It has the following structural formula:
C 25H 27CIN 2·2HCl·H 20 M.W. 481.88544 g/mol
-
CLINICAL PHARMACOLOGY
Meclizine Hydrochloride, USP is an antihistamine that shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
Pharmacokinetics
The available pharmacokinetic information for Meclizine Hydrochloride, USP following oral administration has been summarized from published literature.
Absorption
Meclizine Hydrochloride, USP is absorbed after oral administration with maximum plasma concentrations reaching at a median T max value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Distribution
Drug distribution characteristics for Meclizine Hydrochloride, USP in humans remains unknown.
Metabolism
The metabolic fate of Meclizine Hydrochloride, USP in humans is unknown. In an in vitro metabolic study using human hepatic microsome and recombinant CYP enzyme, CYP 2D6 was found to be the dominant enzyme for metabolism of Meclizine Hydrochloride, USP.
The genetic polymorphism of CYP2D6 that results in extensive-, poor-, intermediate- and ultrarapid metabolizer phenotypes could contribute to large inter-individual variability in Meclizine Hydrochloride, USP exposure.
-
INDICATIONS AND USAGE
For Consumers (the general public):
Naus-Ease® (Meclizine Hydrochloride), USP Film Strips are used for the prevention and treatment of nausea and vomiting, or dizziness associated with motion sickness.For Health Professionals:
Based on a review of Meclizine Hydrochloride, USP drug by the National Academy of Sciences – National Research Council and/or other information, FDA has classified the indications of Meclizine Hydrochloride, USP as follows:
1. The prevention and treatment (management) of nausea and vomiting, and dizziness associated with motion sickness.
2. For the treatment of vertigo. - CONTRAINDICATIONS
-
WARNINGS
Clinical studies establishing safety and effectiveness in children under 12 years of age have not been done; therefore, usage is not recommended in children under 12 years of age unless directed by a doctor.
Due to its potential anticholinergic action, do not take unless directed by a doctor if you have a breathing problem such as asthma, emphysema, or chronic bronchitis, glaucoma, or difficulty in urination due to enlargement of the prostate gland.
Ask a doctor before use if you are taking sedatives or tranquilizers.
When using this product:
1. Do not exceed the recommended dosage.
2. May cause drowsiness.
3. Patients should avoid alcoholic beverages while taking this drug.
4. Since drowsiness may, on occasion, occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery. Therefore patients are reminded caution when driving or operating machinery.
5. Alcohol, sedatives and tranquilizers may increase drowsiness.If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, seek medical help or contact a Poison Control Center right away.
Call Poison Control at 1-800-222-1222 -
PRECAUTIONS
Pediatric Use
Clinical studies establishing safety and effectiveness in children under 12 years of age have not been done; therefore, usage is not recommended in children under 12 years of age unless directed by a doctor.
Pregnancy Use
Pregnancy Category B. Reproduction studies in rats have shown cleft palates at 25 to 50 times the human dose. Epidemiological studies in pregnant women, however, do not indicate that Meclizine Hydrochloride, USP increases the risk of abnormalities when administered during pregnancy. Despite the animal findings, it would appear that the possibility of fetal harm is remote. Nevertheless, Meclizine Hydrochloride, USP or any other medication, should be used during pregnancy only if clearly necessary, and after speaking with a health professional.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Meclizine Hydrochloride, USP is administered to a nursing woman.
Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of meclizine has not been evaluated. As Meclizine Hydrochloride, USP undergoes metabolism, hepatic impairment may result in increased systemic exposure of the drug. Treatment with Meclizine Hydrochloride, USP should be administered with caution in patients with hepatic impairment.
Renal Impairment
The effect of renal impairment on the pharmacokinetics of Meclizine Hydrochloride, USP has not been evaluated. Due to a potential for drug/metabolite accumulation, Meclizine Hydrochloride, USP should be administered with caution in patients with renal impairment and in the elderly as renal function generally declines with age.
Drug Interactions
There may be increased CNS depression when Meclizine Hydrochloride, USP is administered concurrently with other CNS depressants, including alcohol, tranquilizers and sedatives. (see WARNINGS)
Based on in vitro evaluation, Meclizine Hydrochloride, USP is metabolized by CYP2D6. Therefore there is a possibility for a drug interaction between Meclizine Hydrochloride, USP and CYP2D6 inhibitors.
- KEEP OUT OF REACH OF CHILDREN
- ADVERSE REACTIONS
-
DOSAGE AND ADMINISTRATION (DIRECTIONS)
Adults and children 12 years of age and over: Take 1 to 2 Naus-Ease® (Meclizine Hydrochloride), USP Film Strips (25 mg to 50 mg) once daily, or as directed by a physician. Place a film strip on the tongue. Allow each film strip to completely dissolve, and then swallow.
The initial dose of 25 mg to 50 mg of Naus-Ease® (Meclizine Hydrochloride), USP Film Strips should be taken one hour prior to travel for the prevention and treatment of motion sickness. Thereafter, the dose may be repeated every 24 hours for the duration of the journey.
- INACTIVE INGREDIENTS
-
HOW SUPPLIED
Naus-Ease® (Meclizine Hydrochloride), USP Film Strips are available in 25mg strengths. Each Film Strip is individualy wrapped in a pouch, and is available in 8 and 16 count package sizes.
25 mg (Green rectangular shaped, with "S1" printed on each film strip)
Naus-Ease® (Meclizine Hydrochloride), USP; 8 Film Strips: NDC 49467-104-01
Naus-Ease® (Meclizine Hydrochloride), USP; 16 Film Strips: NDC 49467-104-16Distributed by:
Sunascen Therapeutics LLC
Rockville, MD 20850 USA - QUESTIONS OR COMMENTS?
-
PRINCIPAL DISPLAY PANEL
Naus-Ease® (Meclizine Hydrochloride), USP; Film Strips 25 mg Each
Antiemetic – Prevent and Treat: Nausea and VomitingTAMPER EVIDENT: EACH FILM STRIP IS INDIVIDUALLY SEALED IN A POUCH. DO NOT USE IF INDIVIDUAL POUCH IS TORN, BROKEN, OR SHOWS ANY SIGNS OF TAMPERING.
Note:
Store at controlled room temperature 20-30°C. (68-86°F)
Protect from heat and humidity
Use by the expiration date on the package -
INGREDIENTS AND APPEARANCE
NAUS-EASE
meclizine hydrochloride film, solubleProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49467-104 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MECLIZINE HYDROCHLORIDE (UNII: HDP7W44CIO) (MECLIZINE - UNII:3L5TQ84570) MECLIZINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE OXIDE 200000 (UNII: 11628IH70O) HYPROMELLOSES (UNII: 3NXW29V3WO) MALTITOL (UNII: D65DG142WK) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) ANHYDROUS TRISODIUM CITRATE (UNII: RS7A450LGA) Product Characteristics Color green Score no score Shape RECTANGLE Size 22mm Flavor LIME Imprint Code S1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49467-104-01 8 in 1 CARTON 04/16/2012 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:49467-104-16 16 in 1 CARTON 04/16/2012 2 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part336 04/16/2012 Labeler - Sunascen Therapeutics LLC (078272834) Registrant - Sunascen Therapeutics LLC (078272834) Establishment Name Address ID/FEI Business Operations Sunascen Therapeutics LLC 078272834 label(49467-104)