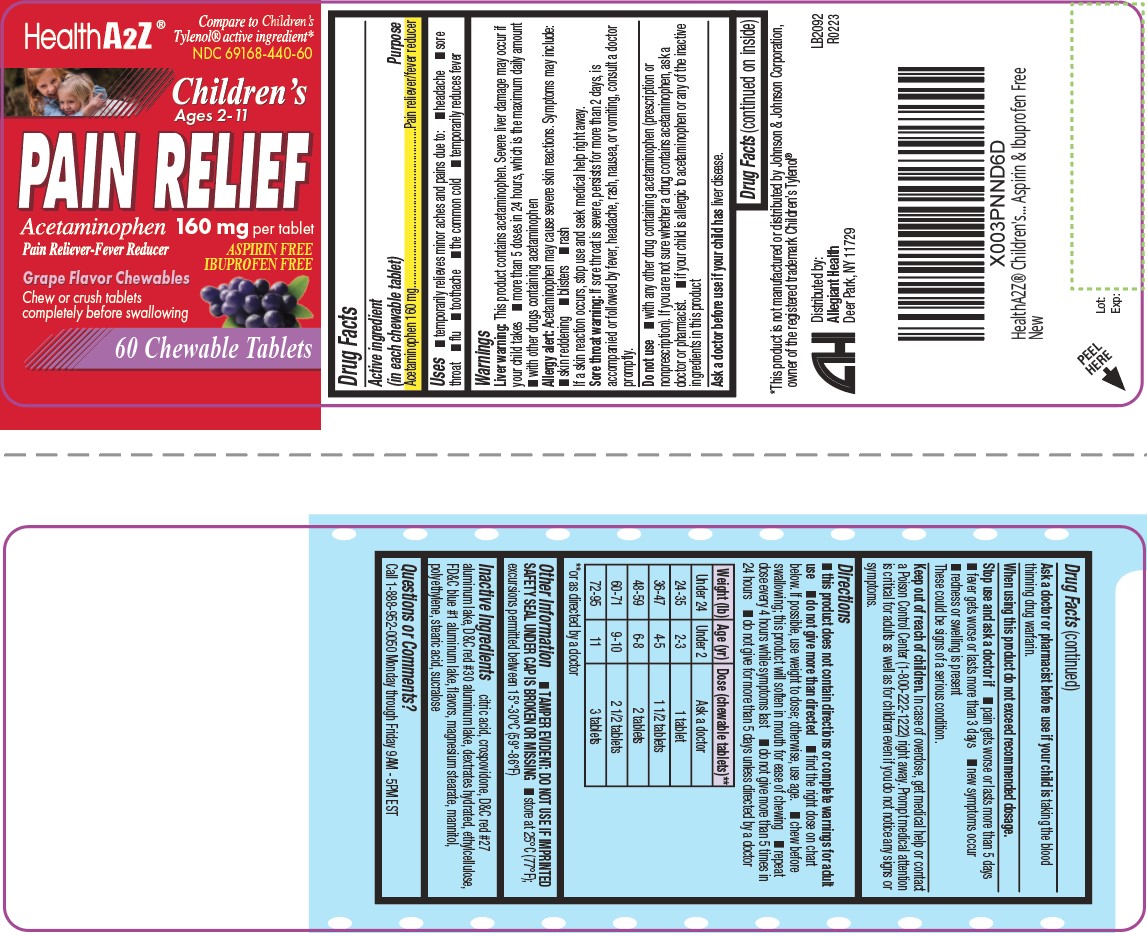
Label: CHILDREN PAIN RELIEF tablet, chewable
- NDC Code(s): 69168-440-60
- Packager: Allegiant Health
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated July 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient(s)
- Purpose
- Use(s)
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your
child takes- more than 5 doses in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or
followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or
nonprescription). If you are not sure whether a drug contains acetaminophen, ask a
doctor or pharmacist. - if your child is allergic to acetaminophen or any of the inactive
ingredients in this product
-
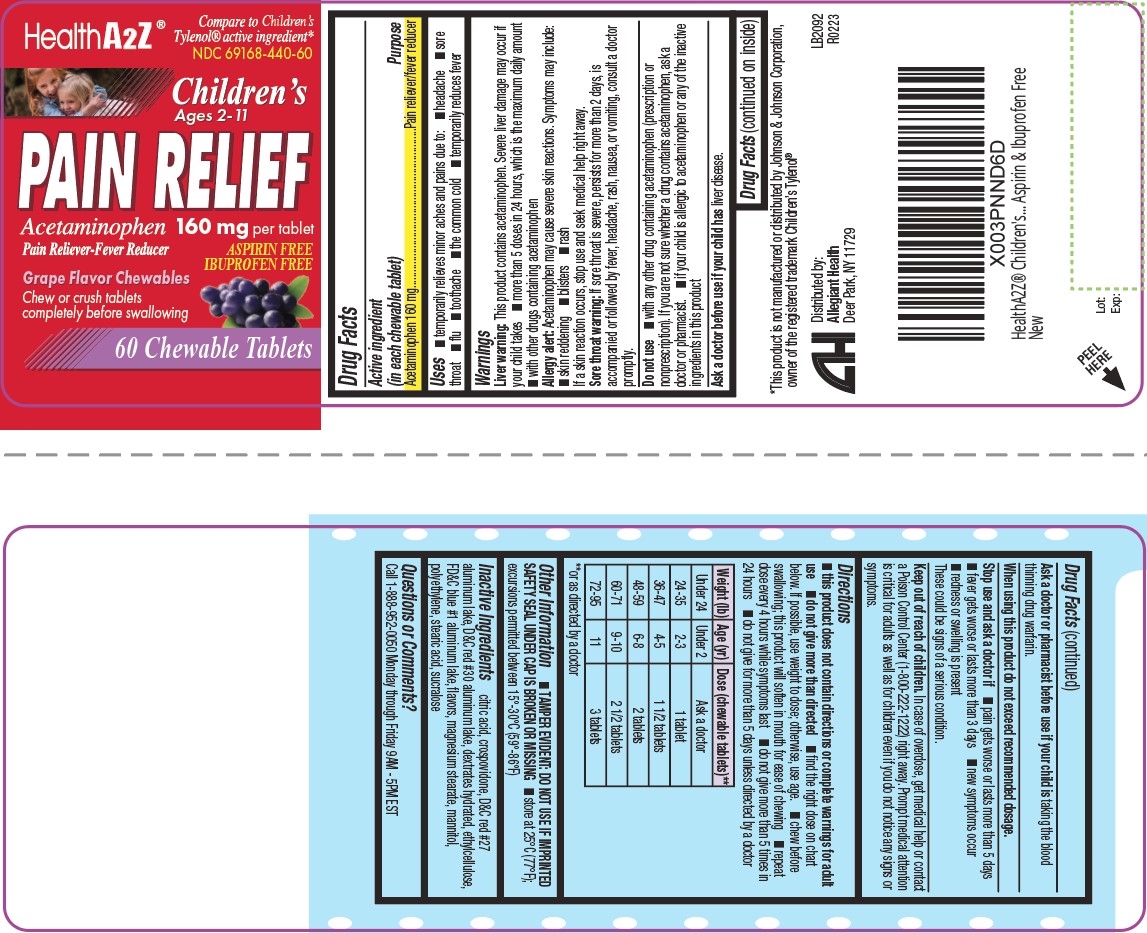
Directions
this product does not contain directions or complete warnings for adult
use- do not give more than directed
- find the right dose on chart below. If possible, use weight to dose; otherwise, use age.
- chew before swallowing; this product will soften in mouth for ease of chewing
- repeat dose every 4 hours while symptoms last
- do not give more than 5 times in 24 hours
- do not give for more than 5 days unless directed by a doctor
Weight (lb) Age (yr) Dose (chewable tablets)** Under 24
Under 2
Ask a doctor
24-35
2-3
1 tablet
36-47
4-5
1 1/2 tablets
48-59
6-8
2 tablets
60-71
9-10
2 1/2 tablets
72-95
11
3 tablets **or as directed by a doctor
- Other information
- Inactive ingredients
- Questions/Comments
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
CHILDREN PAIN RELIEF
children pain relief tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69168-440 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) CROSPOVIDONE (UNII: 2S7830E561) D&C RED NO. 27 ALUMINUM LAKE (UNII: ZK64F7XSTX) D&C RED NO. 30 (UNII: 2S42T2808B) DEXTRATES (UNII: G263MI44RU) ETHYLCELLULOSE, UNSPECIFIED (UNII: 7Z8S9VYZ4B) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) STEARIC ACID (UNII: 4ELV7Z65AP) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color purple Score 2 pieces Shape ROUND Size 16mm Flavor GRAPE Imprint Code 44;449 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69168-440-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 07/18/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 07/18/2023 Labeler - Allegiant Health (079501930)