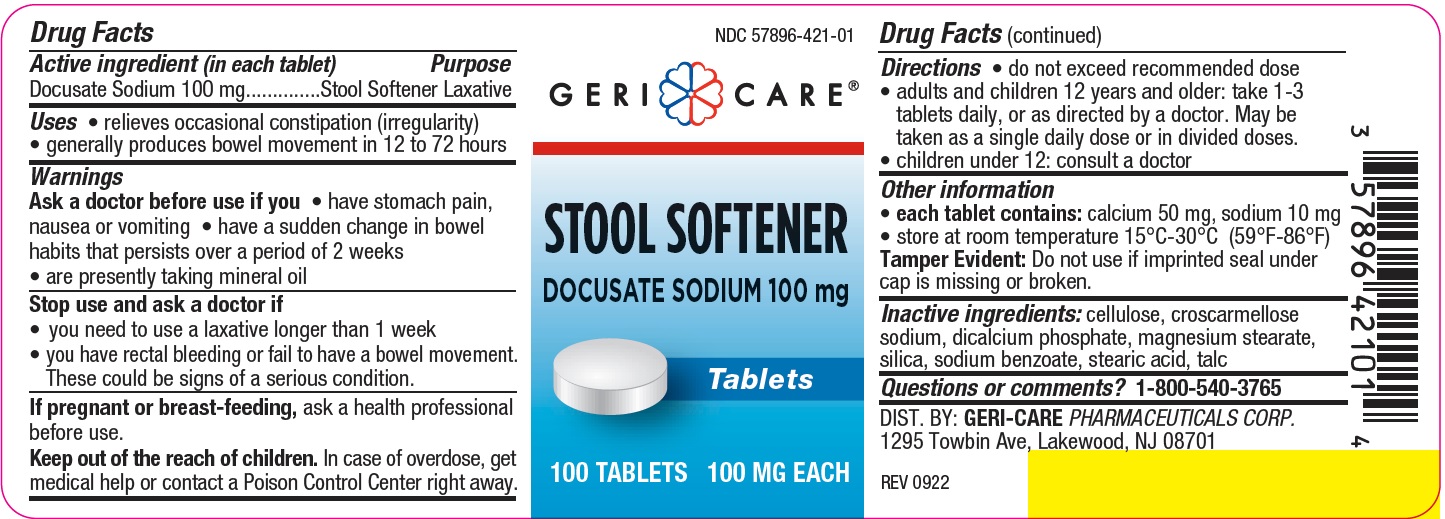
Label: STOOL SOFTENER- docusate sodium tablet
- NDC Code(s): 57896-421-01, 57896-421-10, 57896-421-20
- Packager: Geri-Care Pharmaceuticals, Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you
- have stomach pain, nausea or vomiting
- have a sudden change in bowel habits that persists over a period of 2 weeks
- are presently taking mineral oil
Stop use and ask a doctor if
- you need to use a laxative longer than 1 week
- you have rectal bleeding or fail to have a bowel movement. These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
- Keep out of reach of children
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
STOOL SOFTENER
docusate sodium tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57896-421 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCUSATE SODIUM (UNII: F05Q2T2JA0) (DOCUSATE - UNII:M7P27195AG) DOCUSATE SODIUM 100 mg Inactive Ingredients Ingredient Name Strength SODIUM BENZOATE (UNII: OJ245FE5EU) STEARIC ACID (UNII: 4ELV7Z65AP) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color white Score no score Shape ROUND Size 11mm Flavor Imprint Code GC422 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57896-421-10 1000 in 1 BOTTLE; Type 0: Not a Combination Product 09/01/2015 2 NDC:57896-421-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 09/01/2015 3 NDC:57896-421-20 200 in 1 BOTTLE; Type 0: Not a Combination Product 05/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 09/01/2015 Labeler - Geri-Care Pharmaceuticals, Corp (611196254) Registrant - GCP Laboratories (965480861) Establishment Name Address ID/FEI Business Operations GCP Laboratories 965480861 manufacture(57896-421)