Label: INSTANT HAND SANITIZER- alcohol gel
- NDC Code(s): 61010-1112-1, 61010-1112-2, 61010-1112-3, 61010-1112-8
- Packager: Safetec of America
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL – 0.9g Pouch Label
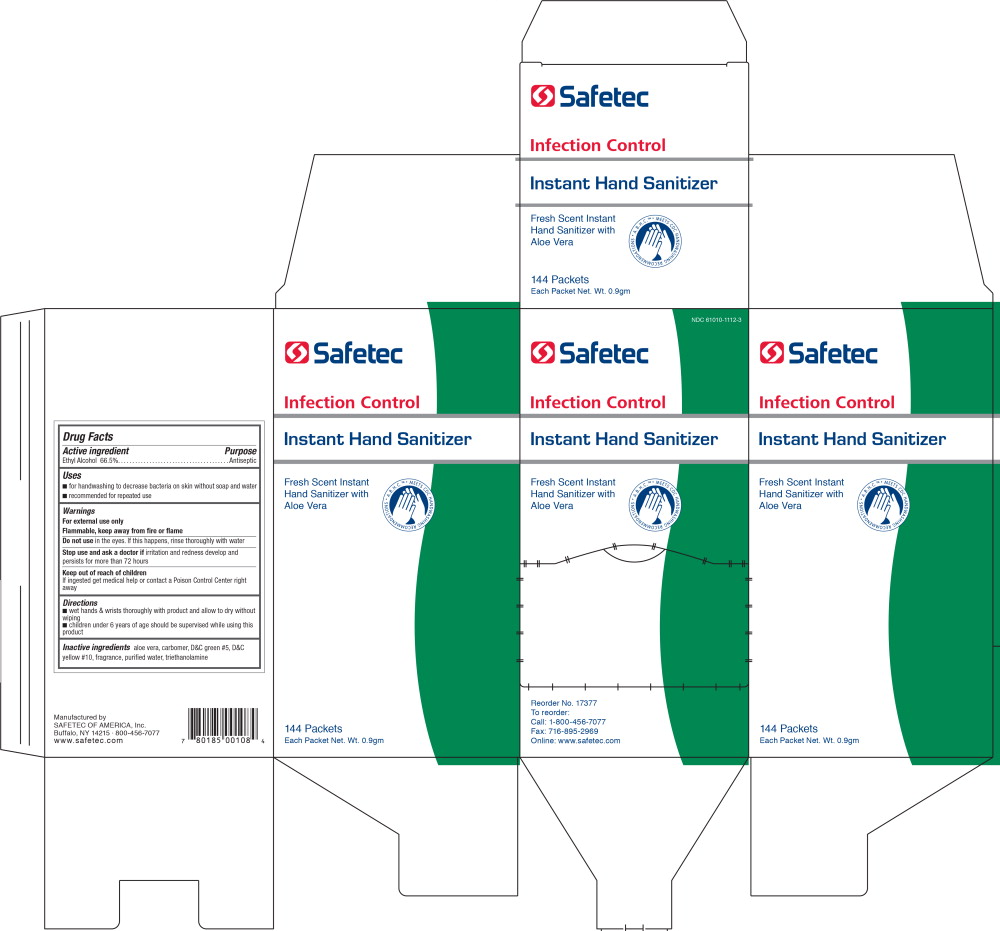
- PRINCIPAL DISPLAY PANEL – 144 Packets Box Label
-
INGREDIENTS AND APPEARANCE
INSTANT HAND SANITIZER
alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61010-1112 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 665 mL in 1 L Inactive Ingredients Ingredient Name Strength ALOE (UNII: V5VD430YW9) WATER (UNII: 059QF0KO0R) TROLAMINE (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61010-1112-2 25 in 1 BOX 12/15/2010 1 NDC:61010-1112-1 0.034 L in 1 POUCH; Type 0: Not a Combination Product 2 NDC:61010-1112-3 144 in 1 BOX 12/15/2010 2 NDC:61010-1112-1 0.034 L in 1 POUCH; Type 0: Not a Combination Product 3 NDC:61010-1112-8 10 in 1 BOX 04/01/2020 3 NDC:61010-1112-1 0.034 L in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 12/15/2010 Labeler - Safetec of America (874965262) Establishment Name Address ID/FEI Business Operations Safetec of America, Inc. 874965262 manufacture(61010-1112)