Label: TRI-VITAMIN WITH FLUORIDE- vitamin a, ascorbic acid, vitamin d, and sodium fluoride solution/ drops
- NHRIC Code(s): 44946-1036-8
- Packager: Sancilio & Company Inc
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated December 27, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- HEALTH CLAIM
- DIRECTIONS
- STATEMENT OF IDENTITY
-
Storage
Store in a cool, dry place at room temperature 20°-25°C (68°-77°F) away from heat and sunlight. Store in original container.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN. IF OVERDOSAGE IS SUSPECTED, SEEK PROFESSIONAL ASSISTANCE OR CONTACT A POISON CONTROL CENTER IMMEDIATELY 1-800-222-1222.
-
Caution
Do not use this product if you are allergic to any of the ingredients. Take this product at least 2 hours before or after taking any products containing calcium (including milk, yogurt, other dairy products) or aluminum/magnesium hydroxide (e.g., certain antacids/laxatives) since these may decrease effectiveness.
Prolonged daily ingestion of excessive fluoride may result in varying degrees of dental fluorosis.
- HEALTH CLAIM
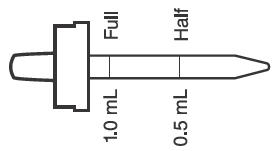
- PRINCIPAL DISPLAY PANEL - 50 mL Bottle Label
-
INGREDIENTS AND APPEARANCE
TRI-VITAMIN WITH FLUORIDE
vitamin a, ascorbic acid, vitamin d, and sodium fluoride solution/ dropsProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:44946-1036 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A (UNII: 81G40H8B0T) (Vitamin A - UNII:81G40H8B0T) Vitamin A 1500 [iU] in 1 mL Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 35 mg in 1 mL Vitamin D (UNII: 9VU1KI44GP) (Vitamin D - UNII:9VU1KI44GP) Vitamin D 400 [iU] in 1 mL Sodium Fluoride (UNII: 8ZYQ1474W7) (Fluoride Ion - UNII:Q80VPU408O) Fluoride Ion 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Polysorbate 80 (UNII: 6OZP39ZG8H) Sodium hydroxide (UNII: 55X04QC32I) Propylene glycol (UNII: 6DC9Q167V3) Sucralose (UNII: 96K6UQ3ZD4) Methylparaben (UNII: A2I8C7HI9T) Propylparaben (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:44946-1036-8 50 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Dietary Supplement 12/20/2011 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color flavor Labeler - Sancilio & Company Inc (176681257)