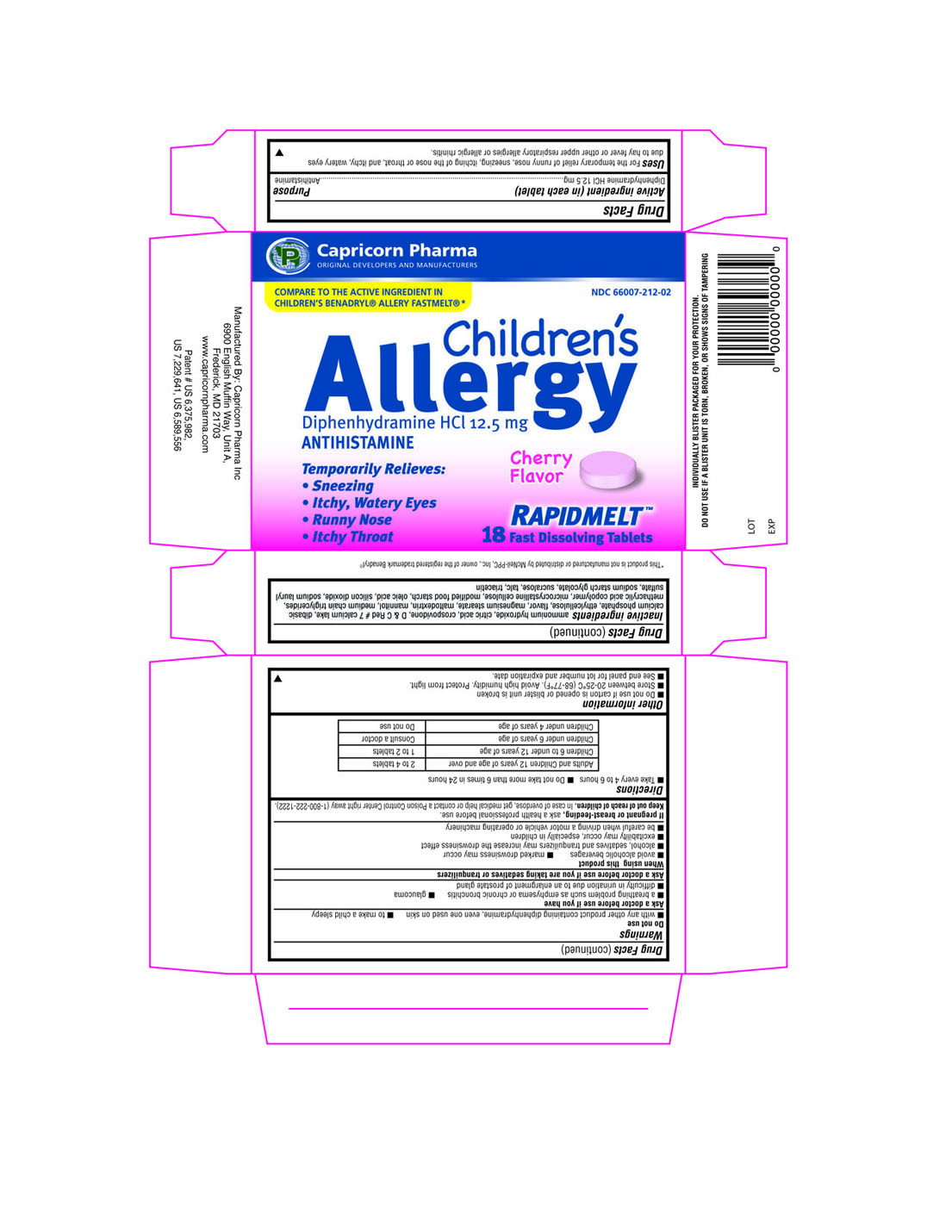
Label: CHILDRENS ALLERGY- diphenhydramine hydrochloride tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 66007-212-01, 66007-212-02 - Packager: Capricorn Pharma Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 4, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- INDICATIONS & USAGE
-
Warnings
Do not use
- with any other product containing Diphenhydramine, even one used on skin
- to make a child sleepy
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to an enlargement of prostate gland
- Directions
- Other Information
-
Inactive ingredients
aqueous ethyl cellulose dispersion, citric acid, crospovidone, D and C Red # 7 calcium lake, dibasic calcium phosphate, flavor, magnesium stearate, maltodextrin, mannitol, methacrylic acid copolymer, microcrystalline cellulose, modified food starch, silicon dioxide, sodium lauryl sulfate, sodium starch glycolate, sucralose, talc, triacetin
-
Principal Display Panel
Capricorn Pharma
ORIGINAL DEVELOPERS AND MANUFACTURERS
NDC 66007-212-02
COMPARE TO THE ACTIVE INGREDIENT IN CHILDREN'S BENADRYL ALLERGY FASTMELT
Children's Allergy
Diphenhydramine HCl 12.5mg
ANTIHISTAMINE
Temporarily Relieves:
- Sneezing
- Itchy, Watery Eyes
- Runny Nose
- Itchy Throat
RAPIDMELT
18 Fast Dissolving Tablets
-
INGREDIENTS AND APPEARANCE
CHILDRENS ALLERGY
diphenhydramine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66007-212 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg Inactive Ingredients Ingredient Name Strength ETHYLCELLULOSE (UNII: 7Z8S9VYZ4B) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) CROSPOVIDONE (UNII: 68401960MK) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MANNITOL (UNII: 3OWL53L36A) METHACRYLIC ACID - ETHYL ACRYLATE COPOLYMER (1:1) TYPE A (UNII: NX76LV5T8J) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) MODIFIED CORN STARCH (1-OCTENYL SUCCINIC ANHYDRIDE) (UNII: 461P5CJN6T) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) SUCRALOSE (UNII: 96K6UQ3ZD4) TALC (UNII: 7SEV7J4R1U) TRIACETIN (UNII: XHX3C3X673) AMMONIA (UNII: 5138Q19F1X) CAPRYLIC/CAPRIC MONO/DIGLYCERIDES (UNII: U72Q2I8C85) OLEIC ACID (UNII: 2UMI9U37CP) Product Characteristics Color pink Score no score Shape ROUND Size 12mm Flavor CHERRY Imprint Code None Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66007-212-02 3 in 1 CARTON 1 NDC:66007-212-01 6 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 09/29/2010 Labeler - Capricorn Pharma Inc. (041704524) Registrant - Capricorn Pharma Inc. (041704524) Establishment Name Address ID/FEI Business Operations Capricorn Pharma Inc. 041704524 manufacture