Label: PRENATAL- vitamin a palmitate, ascorbic acid, cholecalciferol, .alpha.-tocopherol acetate, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, calcium carbonate, ferrous fumarate, and zinc oxide tablet, coated
- NHRIC Code(s): 76413-325-01
- Packager: Central Texas Community Health Centers
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated September 29, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SAFE HANDLING WARNING
-
STATEMENT OF IDENTITY
Supplement Facts Serving Size: 1 Tablet Amount Per Serving % DV* - *
- % Daily Value for Pregnant and Lactating Women
Vitamin A (Palmitate) 4000 IU 50% Vitamin C (Ascorbic Acid) 120 mg 200% Vitamin D (Cholecalciferol) 400 IU 100% Vitamin E (dl - Alpha Tocopheryl Acetate) 30 IU 100% Vitamin B-1 (Thiamine Mononitrate) 1.8 mg 106% Vitamin B-2 (Riboflavin) 1.7 mg 85% Niacin (Niacinamide) 20 mg 100% Vitamin B-6 (Pyridoxine HCl) 2.6 mg 104% Folic Acid 0.8 mg 100% Vitamin B-12 (Cyanocobalamin) 8 mcg 100% Calcium (Calcium Carbonate) 200 mg 15% Iron (Ferrous Fumarate) 28 mg 156% Zinc (Oxide) 25 mg 167% Other ingredients: cellulose, croscarmellose sodium, magnesium stearate, hypromellose, PEG, mineral oil, titanium dioxide, talc, FD&C Red #40, FD&C Yellow #6, FD&C Blue #2
- INDICATIONS
- DIRECTIONS
- WARNINGS
- HEALTH CLAIM
-
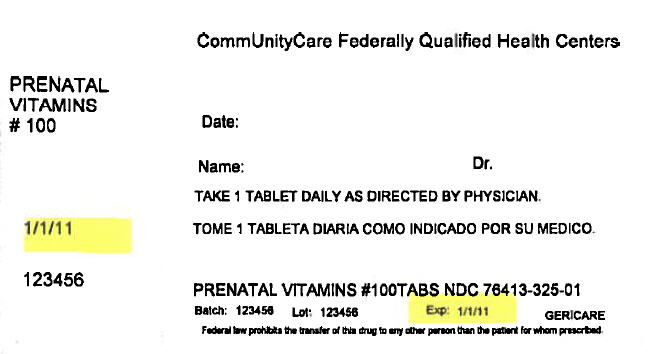
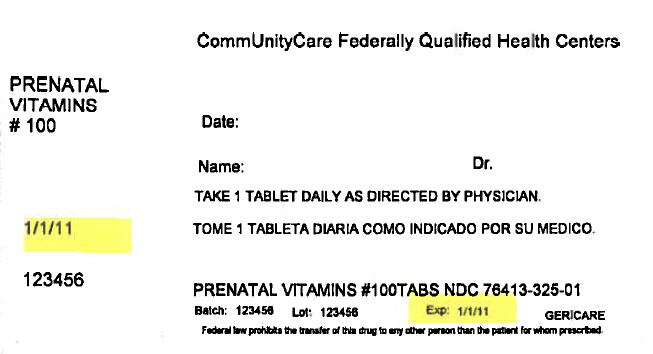
PRINCIPAL DISPLAY PANEL - 100 Tablet Bottle Label
CommUnityCare Federally Qualified Health Centers
PRENATAL
VITAMINS
# 100Date:
Name:
Dr.TAKE 1 TABLET DAILY AS DIRECTED BY PHYSICIAN.
1/1/11
123456
PRENATAL VITAMINS #100TABS NDC 76413-325-01
Batch:
123456
Lot:
123456
Exp:
1/1/11GERICARE
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
-
INGREDIENTS AND APPEARANCE
PRENATAL
vitamin a palmitate, ascorbic acid, cholecalciferol, .alpha.-tocopherol acetate, thiamine mononitrate, riboflavin, niacinamide, pyridoxine hydrochloride, folic acid, cyanocobalamin, calcium carbonate, ferrous fumarate, and zinc oxide tablet, coatedProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:76413-325 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A Palmitate (UNII: 1D1K0N0VVC) (Vitamin A - UNII:81G40H8B0T) Vitamin A 4000 [iU] Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 120 mg Cholecalciferol (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 400 [iU] .Alpha.-Tocopherol Acetate (UNII: 9E8X80D2L0) (.Alpha.-Tocopherol - UNII:H4N855PNZ1) .Alpha.-Tocopherol Acetate 30 [iU] Thiamine Mononitrate (UNII: 8K0I04919X) (Thiamine Ion - UNII:4ABT0J945J) Thiamine 1.8 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 1.7 mg Niacinamide (UNII: 25X51I8RD4) (Niacinamide - UNII:25X51I8RD4) Niacinamide 20 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine 2.6 mg Folic acid (UNII: 935E97BOY8) (Folic acid - UNII:935E97BOY8) Folic acid 0.8 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 8 ug Calcium Carbonate (UNII: H0G9379FGK) (Calcium Cation - UNII:2M83C4R6ZB, Carbonate Ion - UNII:7UJQ5OPE7D) Calcium Carbonate 200 mg Ferrous fumarate (UNII: R5L488RY0Q) (Ferrous Cation - UNII:GW89581OWR) Ferrous Cation 28 mg Zinc oxide (UNII: SOI2LOH54Z) (Zinc oxide - UNII:SOI2LOH54Z) Zinc oxide 25 mg Inactive Ingredients Ingredient Name Strength powdered cellulose (UNII: SMD1X3XO9M) croscarmellose sodium (UNII: M28OL1HH48) magnesium stearate (UNII: 70097M6I30) hypromellose, unspecified (UNII: 3NXW29V3WO) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) mineral oil (UNII: T5L8T28FGP) titanium dioxide (UNII: 15FIX9V2JP) talc (UNII: 7SEV7J4R1U) FD&C Red NO. 40 (UNII: WZB9127XOA) FD&C Yellow NO. 6 (UNII: H77VEI93A8) FD&C Blue NO. 2 (UNII: L06K8R7DQK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:76413-325-01 100 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 06/01/2015 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 20 mm Labeler - Central Texas Community Health Centers (079674019) Establishment Name Address ID/FEI Business Operations Central Texas Community Health Centers 079674019 REPACK, RELABEL Establishment Name Address ID/FEI Business Operations Geri-Care Pharmaceuticals 611196254 MANUFACTURE, REPACK