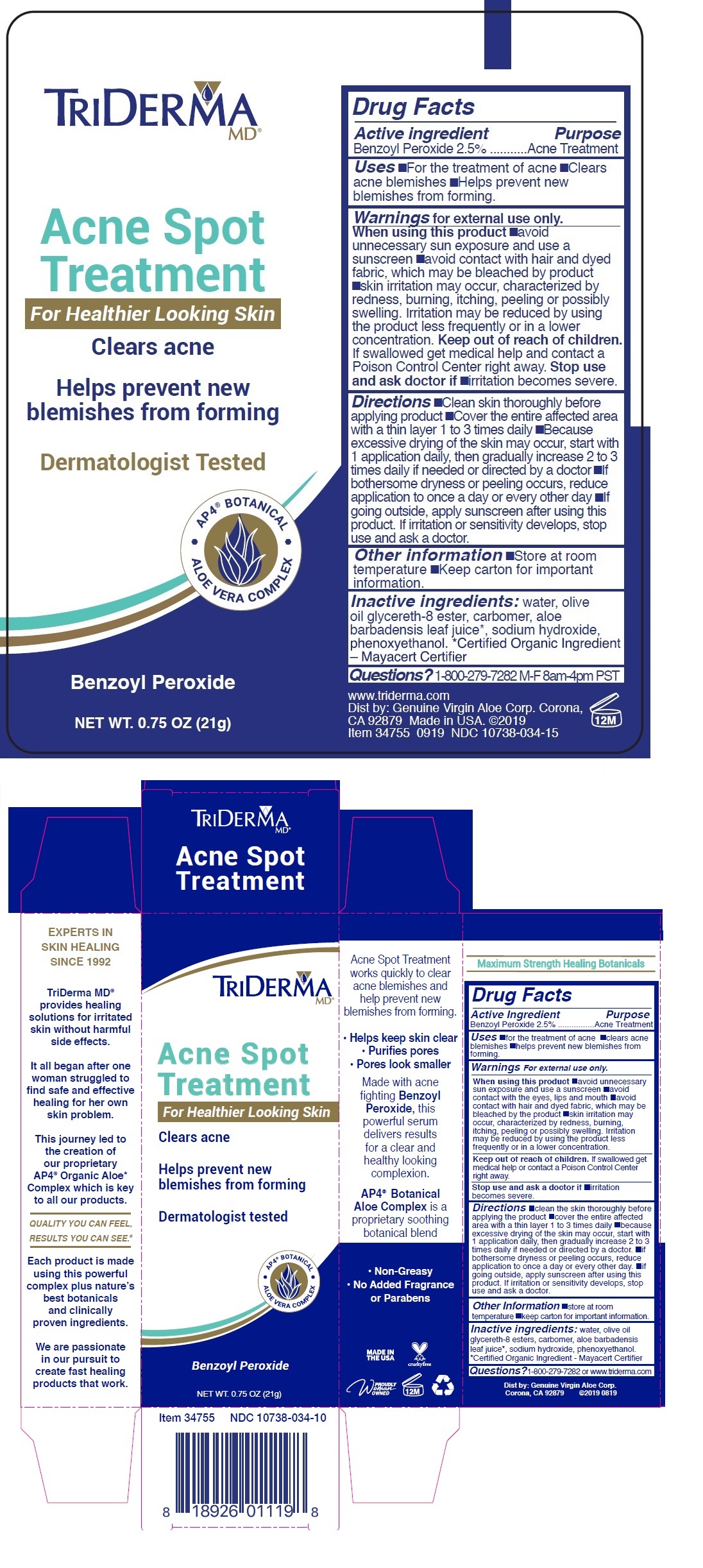
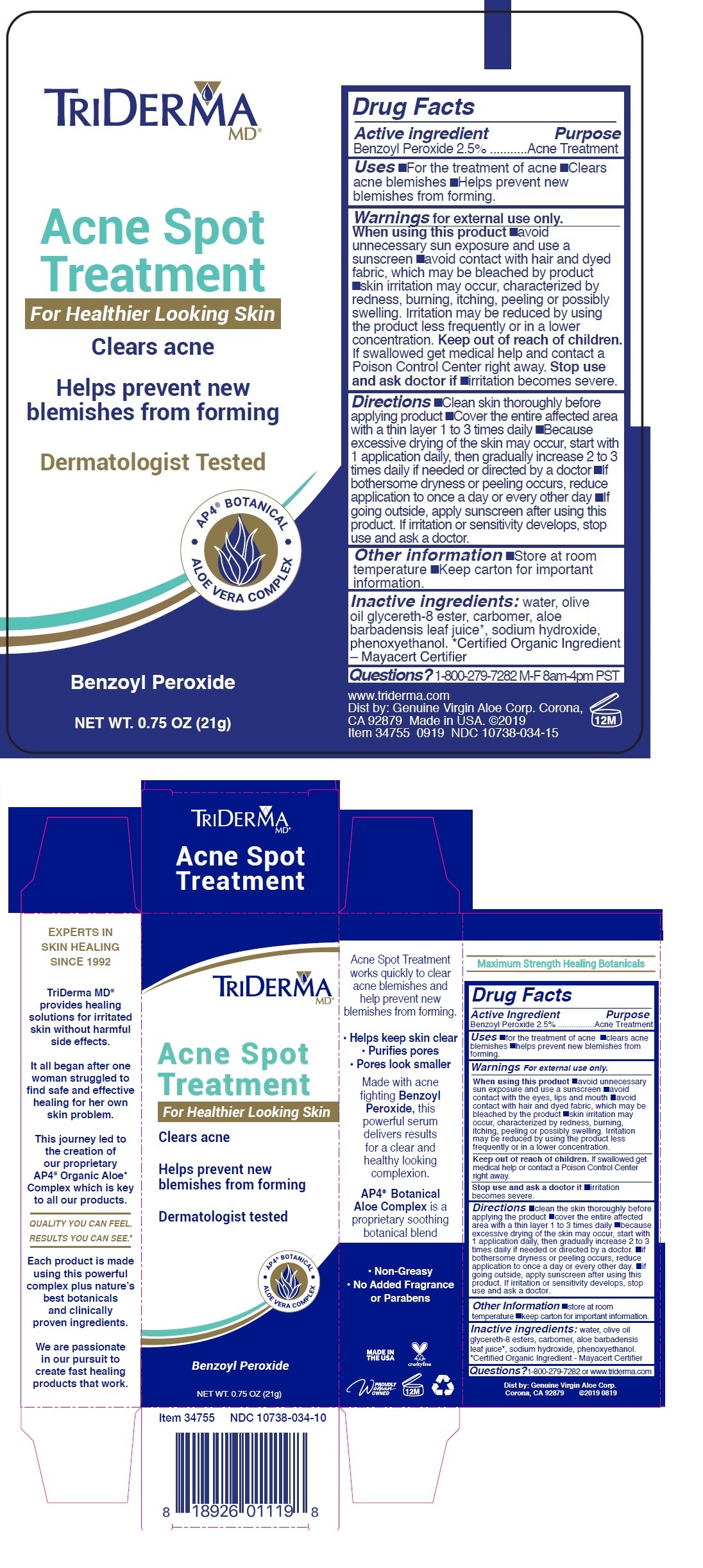
Label: TRIDERMA ACNE SPOT TREATMENT- benzoyl peroxide cream
- NDC Code(s): 10738-034-10, 10738-034-15
- Packager: Genuine Virgin Aloe Corporation
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 2, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- INDICATIONS & USAGE
-
WARNINGS
Warnings For external use only.
When using this product •avoid unnecessary sun exposure and use a sunscreen •avoid contact with the eyes, lips and mouth •avoid contact with hair and dyed fabric, which may be bleached by the product •skin irritation may occur, characterized by redness, burning, itching, peeling or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
Stop use and ask a doctor if •irritation becomes severe.
-
DOSAGE & ADMINISTRATION
Directions •clean the skin thoroughly before applying the product •cover the entire affected area with a thin layer 1 to 3 times daily •because excessive drying of the skin may occur, start with 1 application daily, then gradually increase 2 to 3 times daily if needed or directed by a doctor •if bothersome dryness or peeling occurs, reduce application to once a day or every other day •if going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use and ask a doctor.
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
-
SPL UNCLASSIFIED SECTION
For Healthier Looking Skin
Clears acne
Helps prevent new blemishes from forming
Dermatologist Tested
• AP4 ® BOTANICAL • ALOE VERA COMPLEX
Acne Spot Treatment works quickly to clear acne blemishes and help prevent new blemishes from forming.
• Helps keep skin clear
• Purifies pores
• Pores look smallerMade with acne fighting Benzoyl Peroxide, this powerful serum delivers results for a clear and healthy looking complexion.
AP4 ® Botanical Aloe Complex is a proprietary soothing botanical blend
• Non-Greasy
• No Added Fragrance or ParabensEXPERTS IN SKIN HEALING SINCE 1992
TriDerma MD ® provides healing solutions for irritated skin without harmful side effects.
It all began after one woman struggled to find safe and effective healing for her own skin problem.
This journey led to the creation of our proprietary AP4 ® Organic Aloe* complex which is key to all our products.
QUALITY YOU CAN FEEL, RESULTS YOU CAN SEE ®
Each product is made using this powerful complex plus nature's best botanicals and clinically proven ingredients.
We are passionate in our pursuit to create fast healing products that work.
MADE IN THE USA
Maximum Strength Healing Botanicals
Dist by: Genuine Virgin Aloe Corp. Corona,
CA 92879 ©2019 0819
- packaging
-
INGREDIENTS AND APPEARANCE
TRIDERMA ACNE SPOT TREATMENT
benzoyl peroxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10738-034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 2.5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) OLIVE OIL GLYCERETH-8 ESTERS (UNII: 322K2STO13) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) ALOE VERA LEAF (UNII: ZY81Z83H0X) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10738-034-10 1 in 1 CARTON 12/30/2019 1 NDC:10738-034-15 21 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 12/30/2019 Labeler - Genuine Virgin Aloe Corporation (961374147) Establishment Name Address ID/FEI Business Operations Genuine Virgin Aloe Corporation 961374147 manufacture(10738-034)