Label: BLOOD CLOTTING FIRST AID- benzethonium chloride and lidocaine aerosol, spray
- NDC Code(s): 47682-226-17
- Packager: Unifirst First Aid Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
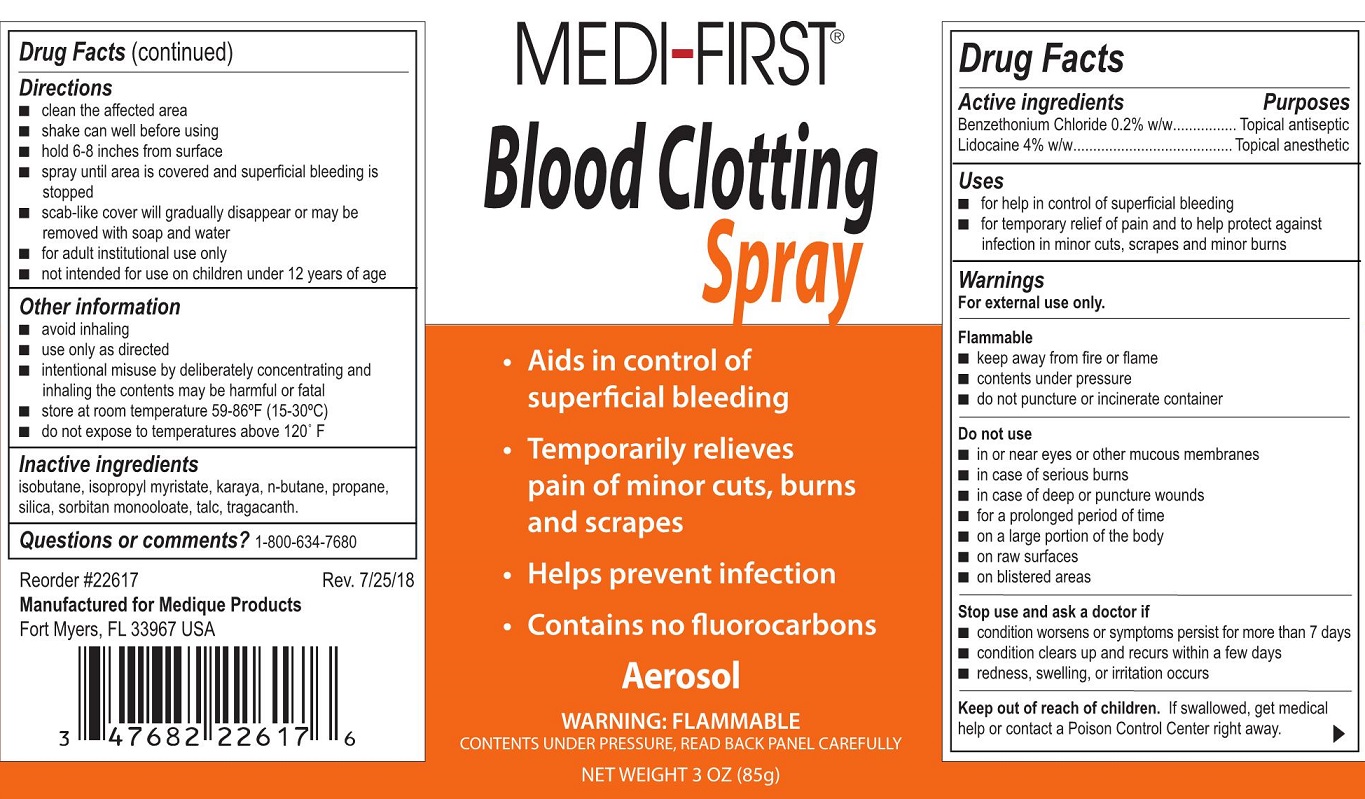
- Drug Facts
- Active Ingredients
- Purpose
- Uses
- Warnings
- Do not use
- Stop use and ask a doctor if
- Keep out of reach of children. .
-
Directions
- clean the affected area
- shake can well before using
- hold 6-8 inches from surface
- spray until area is covered and superficial bleeding is stopped
- scab-like cover will gradually disappear or may be removed with soap and water
- for adult institutional use only
- not intended for use on children under 12 years of age
- Other information
- Inactive ingredients
- Questions or comments?
- Medi-First Blood Clotting Spray Label
-
INGREDIENTS AND APPEARANCE
BLOOD CLOTTING FIRST AID
benzethonium chloride and lidocaine aerosol, sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:47682-226 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 3400 mg in 85 g BENZETHONIUM CHLORIDE (UNII: PH41D05744) (BENZETHONIUM - UNII:1VU15B70BP) BENZETHONIUM CHLORIDE 170 mg in 85 g Inactive Ingredients Ingredient Name Strength ISOBUTANE (UNII: BXR49TP611) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) KARAYA GUM (UNII: 73W9IQY50Q) BUTANE (UNII: 6LV4FOR43R) PROPANE (UNII: T75W9911L6) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) TALC (UNII: 7SEV7J4R1U) TRAGACANTH (UNII: 2944357O2O) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:47682-226-17 85 g in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 04/05/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 04/05/2013 Labeler - Unifirst First Aid Corporation (832947092) Establishment Name Address ID/FEI Business Operations Dixon Investments, Inc. 115315822 manufacture(47682-226)