Label: FAMILY CARE COLD AND FLU MULTI SYMPTOM- acetaminophen, dextromethorphan hydrobromide, and phenylephrine hydrochloride liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 65923-627-04 - Packager: United Exchange Corp.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 23, 2015
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
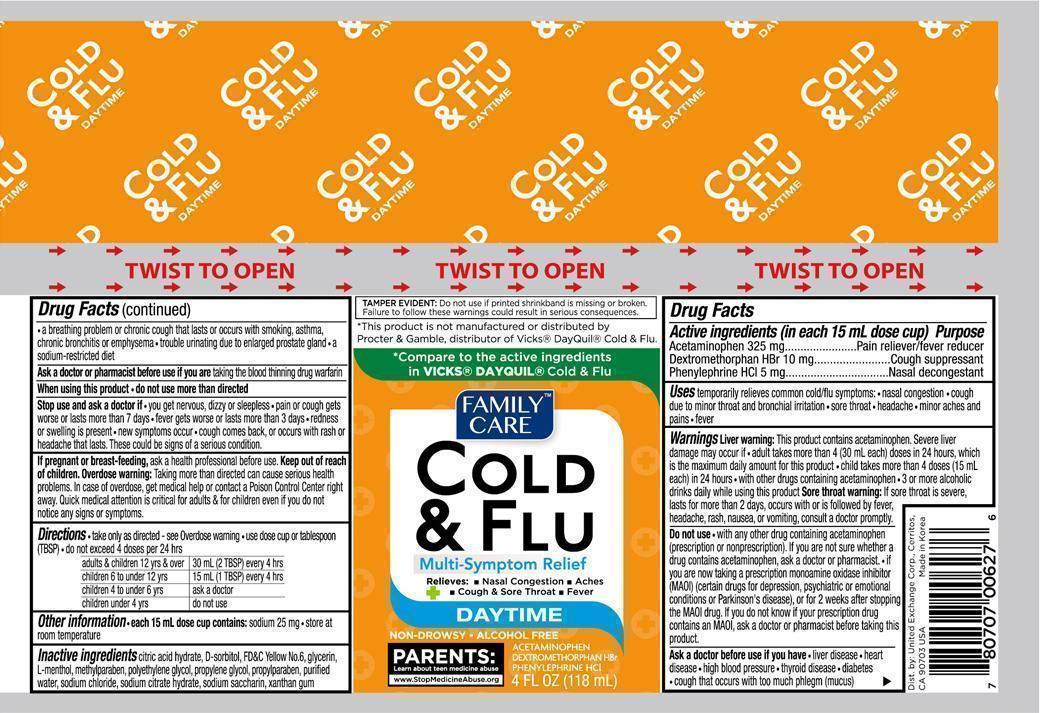
Active ingredients (in each 15 mL dose cup) Purpose
Acetaminophen 325 mg................................................Pain reliever/fever reducer
Dextromethorphan HBr 10 mg..................................................cough suppressant
Phenylephrine HCI 5 mg.........................................................Nasal decongestant
- PURPOSE
-
WARNINGS
Warnings
Liver warning: This product contains acetaminophen. Sever liver damage may occur if
- adult takes more than 4 (30 mL each) doses in 24 hours, which is the maximum daily amount for this product
- child takes more than 4 doses (15 mL each) in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks daily while using this product
Sore throat warning: if sore throat is sever, lasts more than 2 days, occurs with or is followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
-
DO NOT USE
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
-
ASK DOCTOR
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- cough that occurs with too much phlegm (mucus)
- a breathing problem or chronic cough that lasts or occurs with smoking, asthma, chronic bronchitis or emphysema
- trouble urinating due to enlarged prostate gland
- a sodium-resitricted diet
- ASK DOCTOR/PHARMACIST
- WHEN USING
-
STOP USE
Stop use and ask a doctor if
- you get nervous, dizzy or sleepless
- pain or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back, or occurs with rash or headache that lasts. These could be signs of a serious condition.
- PREGNANCY OR BREAST FEEDING
-
KEEP OUT OF REACH OF CHILDREN
Keep out of reach of children. Overdose warning: Taking more than directed can cause serious health problems. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults & for children even if you do not notice any signs or symptoms.
-
INDICATIONS & USAGE
Directions
- take only as directed - see Overdose warning
- use dose cup or tablespoon (TBSP)
- do not exceed 4 doses per 24 hrs
adults & children 12 yrs over: 30 mL (2TBSP) every 4 hours
children 6 to under 12 yrs: 15 mL(1 TBSP) every 4 hours
children 4 to under 6 yrs: ask a doctor
children under 4 yrs: do not use
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FAMILY CARE COLD AND FLU MULTI SYMPTOM
acetaminophen, dextromethorphan hydrobromide, and phenylephrine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65923-627 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg in 15 mL DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg in 15 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg in 15 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SORBITOL (UNII: 506T60A25R) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GLYCERIN (UNII: PDC6A3C0OX) MENTHOL (UNII: L7T10EIP3A) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE (UNII: 1Q73Q2JULR) SACCHARIN SODIUM (UNII: SB8ZUX40TY) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65923-627-04 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/23/2015 Labeler - United Exchange Corp. (840130579)