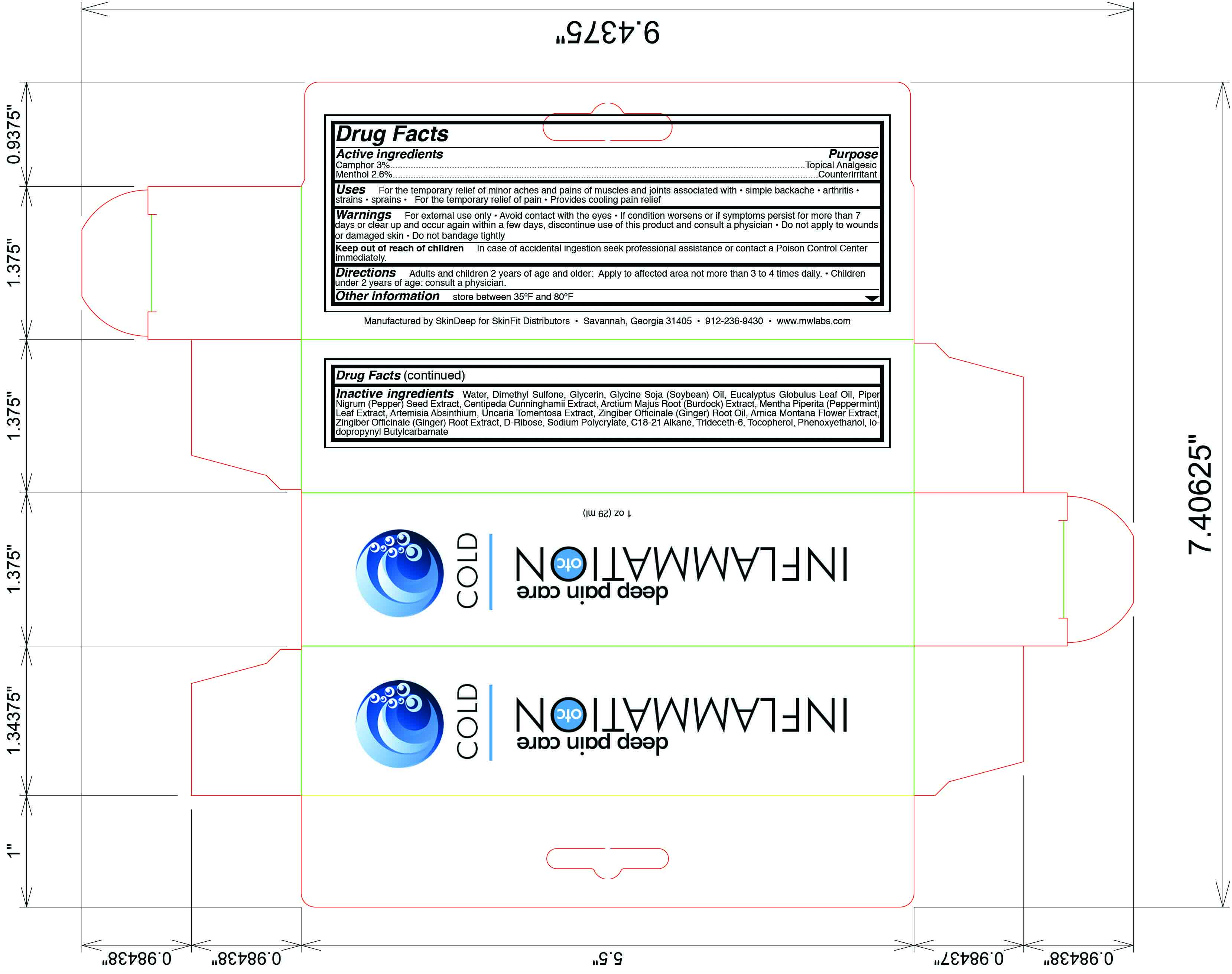
Label: INFLAMMATION OTC DEEP PAIN CARE COLD- camphor, menthol lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 45156-5610-1, 45156-5610-2 - Packager: Skin Deep
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 16, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Keep out of reach of children
- Directions
-
Inactive Ingredients
water, dimethyl sulfone, glycerin, glycine soja (soybean) oil, eucalyptus globulus oil, piper nigrum (pepper) seed extract, centipedia cunninghamii extract, arctium majus root (burdock) extract, mentha piperita (peppermint) leaf extract, artemisia absinthium, uncaria tomentosa extract, zingiber officinale (ginger) root oil, arnica montana flower extract, zingiber officinale (ginger) root extract, d-ribose, sodium polycrylate, c18-21 alkane, trideceth-6, tocopherol, phenoxyethanol, iodopropynyl butylcarbamate
- Questions or Comments
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
INFLAMMATION OTC DEEP PAIN CARE COLD
camphor, menthol lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:45156-5610 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Camphor (Natural) (UNII: N20HL7Q941) (Camphor (Natural) - UNII:N20HL7Q941) Camphor (Natural) 18 g in 600 g Menthol (UNII: L7T10EIP3A) (Menthol - UNII:L7T10EIP3A) Menthol 16 g in 600 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Glycerin (UNII: PDC6A3C0OX) Dimethyl Sulfone (UNII: 9H4PO4Z4FT) Black Pepper Oil (UNII: U17J84S19Z) Soybean Oil (UNII: 241ATL177A) Arctium Lappa Root (UNII: 597E9BI3Z3) Peppermint (UNII: V95R5KMY2B) Wormwood (UNII: F84709P2XV) Cat's Claw (UNII: 9060PRM18Q) Ginger Oil (UNII: SAS9Z1SVUK) Arnica Montana (UNII: O80TY208ZW) Ginger (UNII: C5529G5JPQ) Trideceth-6 (UNII: 3T5PCR2H0C) Alpha-Tocopherol (UNII: H4N855PNZ1) Phenoxyethanol (UNII: HIE492ZZ3T) Iodopropynyl Butylcarbamate (UNII: 603P14DHEB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:45156-5610-2 1 in 1 BOX 1 NDC:45156-5610-1 31 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part348 08/16/2011 Labeler - Skin Deep (833276566) Establishment Name Address ID/FEI Business Operations Skin Deep 833276566 manufacture