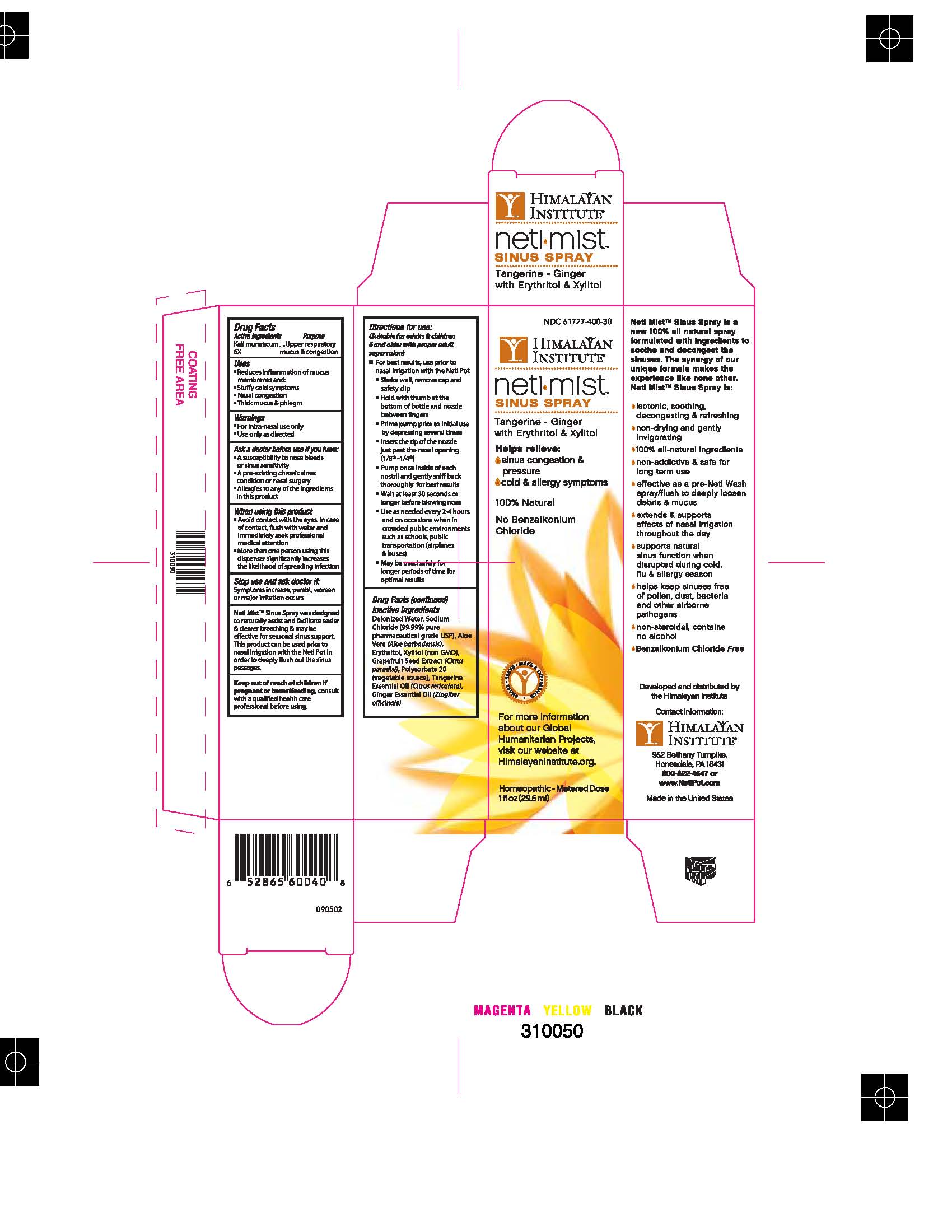
Label: NETI MIST SINUS SPRAY- potassium chloride liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 61727-400-30 - Packager: Homeocare Laboratories
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 14, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Kali muriaticum 6x
Keep out of reach of children if pregnant or breastfeeding, consult with a qualified health care professional before using.
Keep out of reach of children if pregnant or breastfeeding, consult with a qualified health care professional before using.
Inactive Ingredients:
Deionized Water, Sodium Chloride (99.99% pure pharmaceutical grade USP), Aloe Vera (Aloe barbadensis), Erythritol, Xylitol (non GMO), Grapefruit Seed Extract (Citrus paradisi), Polysorbate 20 (vegetable source), Tangerine Essential Oil (Citrus reticulata), Ginger Essential Oil (Zingiber officinale).
-
Neti Mist Sinus Spray
NMSS
NetiMist Sinus Spray.jpg
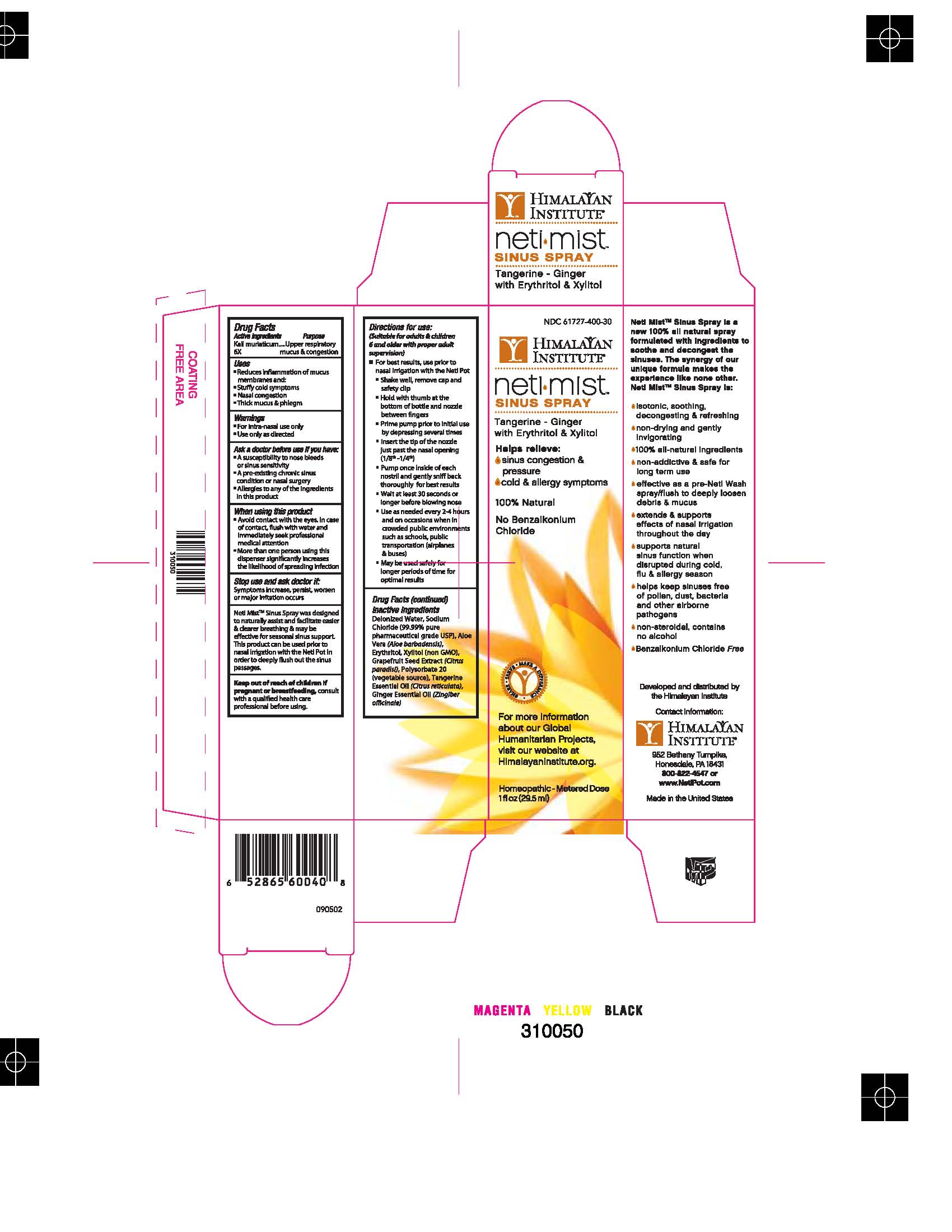

NMSSBOX
BoxProof60040HIM1OZ000.jpg
Dosage & Administration: Directions for Use: (Suitable for adults & children 6 and older with proper adult supervision) *For best results, use prior to nasal irrigation with the Neti Pot • Shake well, remove cap and safely clip • Hold with thumb at the bottom of bottle and nozzle between fingers • Prime pump prior to initial use by depressing several times • Insert the tip of the nozzle just past the nasal opening (1/8th-1/4th) • Pump once inside of each nostril and gently sniff back thoroughly for best results • Wait at least 30 seconds or longer before blowing nose • Use as needed every 2-4 hours and on occasion when in crowded public environments such as schools, public transportation (airplanes & buses) • May be used safely for longer periods of time for optimal results
-
INGREDIENTS AND APPEARANCE
NETI MIST SINUS SPRAY
potassium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:61727-400 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CATION 6 [hp_X] in 29.5 mL Inactive Ingredients Ingredient Name Strength ERYTHRITOL (UNII: RA96B954X6) XYLITOL (UNII: VCQ006KQ1E) CITRUS PARADISI SEED (UNII: 12F08874Y7) MANDARIN OIL (UNII: NJO720F72R) GINGER (UNII: C5529G5JPQ) WATER (UNII: 059QF0KO0R) POLYSORBATE 20 (UNII: 7T1F30V5YH) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61727-400-30 1 in 1 BOX 1 29.5 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/14/2011 Labeler - Homeocare Laboratories (088248828) Registrant - Homeocare Laboratories (088248828) Establishment Name Address ID/FEI Business Operations Homeocare Laboratories 088248828 manufacture