Label: HYDROCORTISONE cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 53210-1002-0 - Packager: Morales Distributors Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 28, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
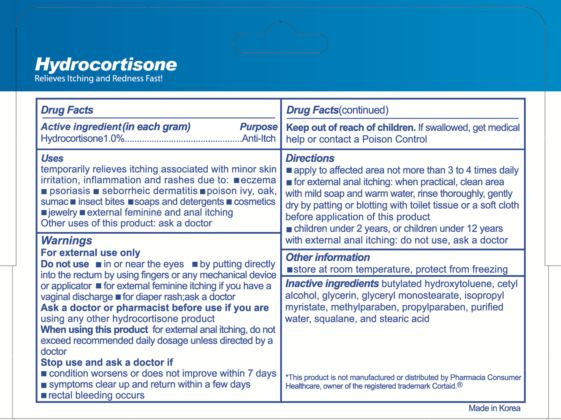
- Active ingredient
- Purpose
- Uses
- Warnings
- Do not use
- Ask a doctor or pharmacist
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- apply to affected area not more than 3 to 4 times daily
- for external anal itching: when practical, clean area with mild soap and warm water, rinse thoroughly, gently dry by patting or blotting with toilet tissue or a soft cloth before application of this product
- children under 2 years, or children under 12 years with external anal itching, do not use, ask a doctor
- Other information
- Inactive ingredients
-
Principal Label
NDC NO. 53210-1002-0
Compare to maximum strength Cortaid(R) active ingredientMaximum Strength
FAST RELIEF
plus itching plus rednessAnti-Itch Cream for Skin Irritations
Relieves Itching and Redness Fast
Hydrocortisone 1 percent Cream
NET 1oz(28.3g)
Exclusively distributed by:
Morales Distributors, Inc
Mayaguez, P.R. 00682This product is not manufactured or distributed by Pharmacia Consumer Healthcare, owner of the registered trademark Cortaid.(R)
Made in Korea


-
INGREDIENTS AND APPEARANCE
HYDROCORTISONE
hydrocortisone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53210-1002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 1 g in 100 g Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SQUALANE (UNII: GW89575KF9) STEARIC ACID (UNII: 4ELV7Z65AP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53210-1002-0 1 in 1 BOX 1 28 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 09/25/2012 Labeler - Morales Distributors Inc (127151731)



