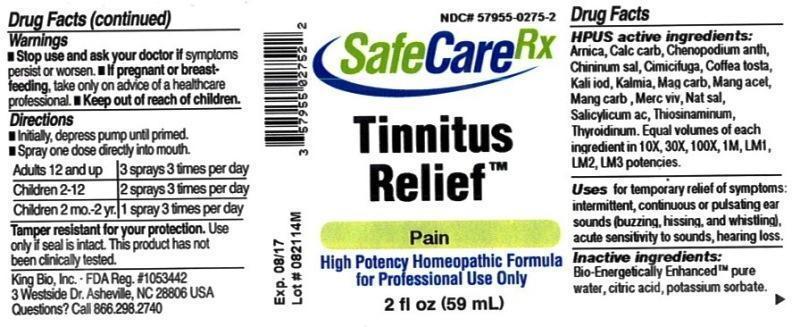
Label: TINNITUS RELIEF- arnica montana, calcarea carbonica, chenopodium anthelminticum, chininum salicylicum, cimicifuga racemosa, coffea tosta, kali iodatum, kalmia latifolia, magnesia carbonica, manganum aceticum, manganum carbonicum, mercurius vivus, natrum salicylicum, salicylicum acidum, thiosinaminum, thyroidinum liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 57955-0275-2 - Packager: King Bio Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 4, 2015
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Drug Facts__________________________________________________________________________________________________________
HPUS active ingredients: Arnica montana, Calcarea carbonica, Chenopodium anthelminticum, Chininum salicylicum, Cimicifuga racemosa, Coffea tosta, Kali iodatum, Kalmia latifolia, Magnesia carbonica, Manganum acxeticum, Manganum carbonicum, Mercurius vivus, Natrum salicylicum, Salicylicum acidum, Thiosinaminum, Thyroidinum. Equal volumes of each ingredient in 10X, 30X, 100X, 1M, LM1, LM2, LM3 potencies.
- INDICATIONS & USAGE
- INACTIVE INGREDIENT
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- PURPOSE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TINNITUS RELIEF
arnica montana, calcarea carbonica, chenopodium anthelminticum, chininum salicylicum, cimicifuga racemosa, coffea tosta, kali iodatum, kalmia latifolia, magnesia carbonica, manganum aceticum, manganum carbonicum, mercurius vivus, natrum salicylicum, salicylicum acidum, thiosinaminum, thyroidinum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57955-0275 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 10 [hp_X] in 59 mL OYSTER SHELL CALCIUM CARBONATE, CRUDE (UNII: 2E32821G6I) (OYSTER SHELL CALCIUM CARBONATE, CRUDE - UNII:2E32821G6I) OYSTER SHELL CALCIUM CARBONATE, CRUDE 10 [hp_X] in 59 mL DYSPHANIA AMBROSIOIDES (UNII: 4H5RSU087I) (CHENOPODIUM AMBROSIOIDES - UNII:4H5RSU087I) DYSPHANIA AMBROSIOIDES 10 [hp_X] in 59 mL QUININE SALICYLATE (UNII: 6DY04L71DR) (SALICYLIC ACID - UNII:O414PZ4LPZ) QUININE SALICYLATE 10 [hp_X] in 59 mL BLACK COHOSH (UNII: K73E24S6X9) (BLACK COHOSH - UNII:K73E24S6X9) BLACK COHOSH 10 [hp_X] in 59 mL COFFEA ARABICA SEED, ROASTED (UNII: 9H58JRT35E) (COFFEA ARABICA SEED, ROASTED - UNII:9H58JRT35E) COFFEA ARABICA SEED, ROASTED 10 [hp_X] in 59 mL POTASSIUM IODIDE (UNII: 1C4QK22F9J) (IODIDE ION - UNII:09G4I6V86Q) IODIDE ION 10 [hp_X] in 59 mL KALMIA LATIFOLIA LEAF (UNII: 79N6542N18) (KALMIA LATIFOLIA LEAF - UNII:79N6542N18) KALMIA LATIFOLIA LEAF 10 [hp_X] in 59 mL MAGNESIUM CARBONATE (UNII: 0E53J927NA) (CARBONATE ION - UNII:7UJQ5OPE7D) MAGNESIUM CARBONATE 10 [hp_X] in 59 mL MANGANESE ACETATE TETRAHYDRATE (UNII: 9TO51D176N) (MANGANESE CATION (2+) - UNII:H6EP7W5457) MANGANESE ACETATE TETRAHYDRATE 10 [hp_X] in 59 mL MANGANESE CARBONATE (UNII: 9ZV57512ZM) (MANGANESE CATION (2+) - UNII:H6EP7W5457) MANGANESE CATION (2+) 10 [hp_X] in 59 mL MERCURY (UNII: FXS1BY2PGL) (MERCURY - UNII:FXS1BY2PGL) MERCURY 10 [hp_X] in 59 mL SODIUM SALICYLATE (UNII: WIQ1H85SYP) (SALICYLIC ACID - UNII:O414PZ4LPZ) SODIUM SALICYLATE 10 [hp_X] in 59 mL SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 10 [hp_X] in 59 mL ALLYLTHIOUREA (UNII: 706IDJ14B7) (ALLYLTHIOUREA - UNII:706IDJ14B7) ALLYLTHIOUREA 10 [hp_X] in 59 mL THYROID, UNSPECIFIED (UNII: 0B4FDL9I6P) (THYROID, UNSPECIFIED - UNII:0B4FDL9I6P) THYROID, UNSPECIFIED 10 [hp_X] in 59 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57955-0275-2 59 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/06/2012 Labeler - King Bio Inc. (617901350) Registrant - King Bio Inc. (617901350) Establishment Name Address ID/FEI Business Operations King Bio Inc. 617901350 manufacture(57955-0275)