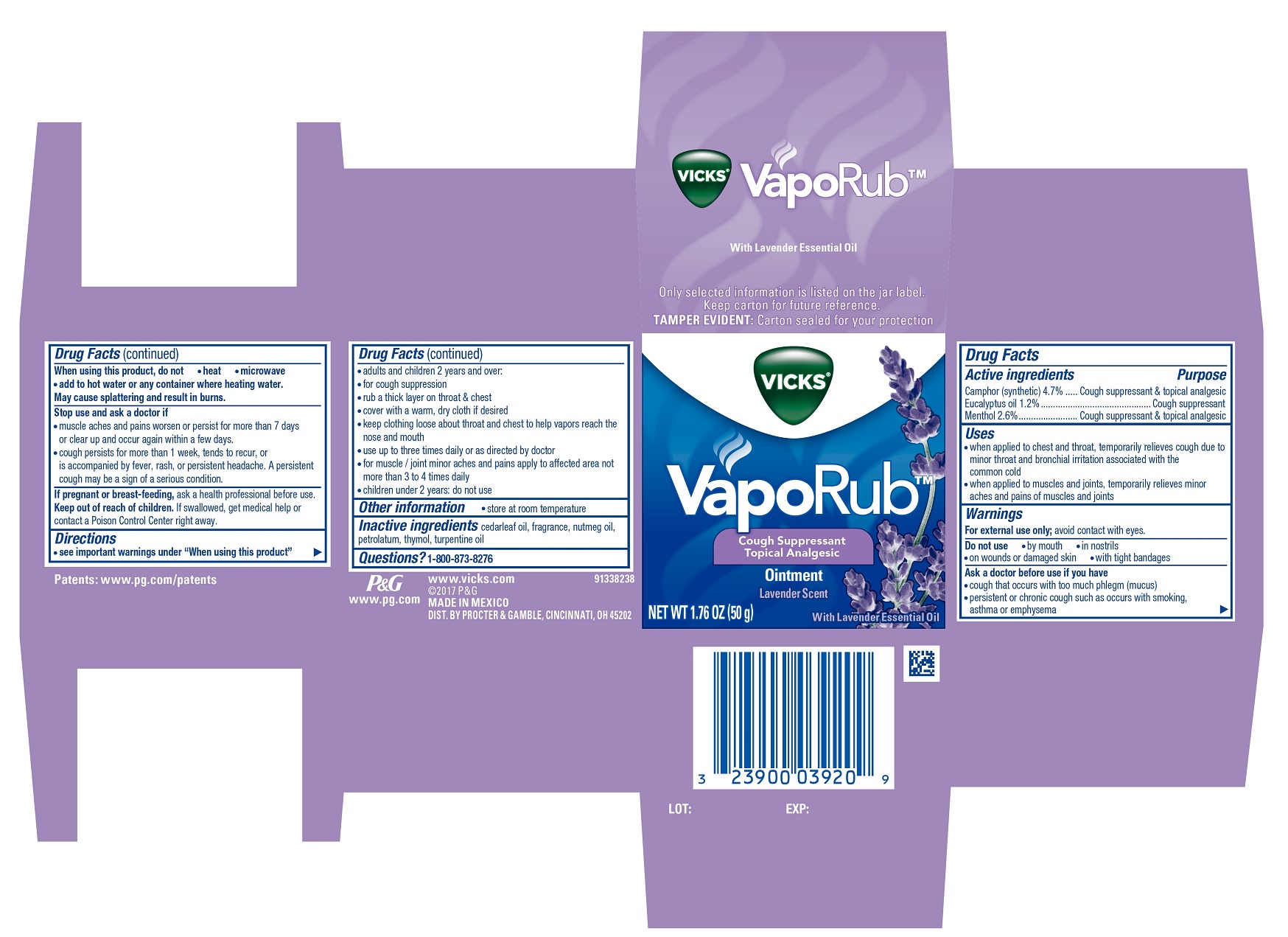
Label: VICKS VAPORUB (camphor- synthetic, eucalyptus oil, menthol ointment
- NDC Code(s): 58933-003-01
- Packager: Procter & Gamble Manufactura S de RL de CV
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Uses
-
Warnings
For external use only;avoid contact with eyes.
Ask a doctor before use if you have
- cough that occurs with too much phlegm (mucus)
- persistent or chronic cough such as occurs with smoking, asthma or emphysema
When using this product, do not
- heat
- microwave
-
add to hot water or any container where heating water.
May cause splattering and result in burns.
Stop use and ask a doctor if
- muscle aches and pains worsen or persist for more than 7 days or clear up and occur again within a few days.
- cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.
-
Directions
- see important warnings under “When using this product”
- adults and children 2 years and over:
- for cough suppression
- rub a thick layer on throat & chest
- cover with a warm, dry cloth if desired
- keep clothing loose about throat and chest to help vapors reach the nose and mouth
- use up to three times daily or as directed by doctor
- for muscle / joint minor aches and pains apply to affected area not more than 3 to 4 times daily
- children under 2 years: do not use
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 50 g Jar Carton
-
INGREDIENTS AND APPEARANCE
VICKS VAPORUB
camphor (synthetic), eucalyptus oil, menthol ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58933-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 0.047 g in 1 g EUCALYPTUS OIL (UNII: 2R04ONI662) (EUCALYPTUS OIL - UNII:2R04ONI662) EUCALYPTUS OIL 0.012 g in 1 g MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.026 g in 1 g Inactive Ingredients Ingredient Name Strength CEDAR LEAF OIL (UNII: BJ169U4NLG) NUTMEG OIL (UNII: Z1CLM48948) THYMOL (UNII: 3J50XA376E) TURPENTINE OIL (UNII: C5H0QJ6V7F) PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58933-003-01 1 in 1 CARTON 04/02/2018 1 50 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/02/2018 Labeler - Procter & Gamble Manufactura S de RL de CV (812807550)