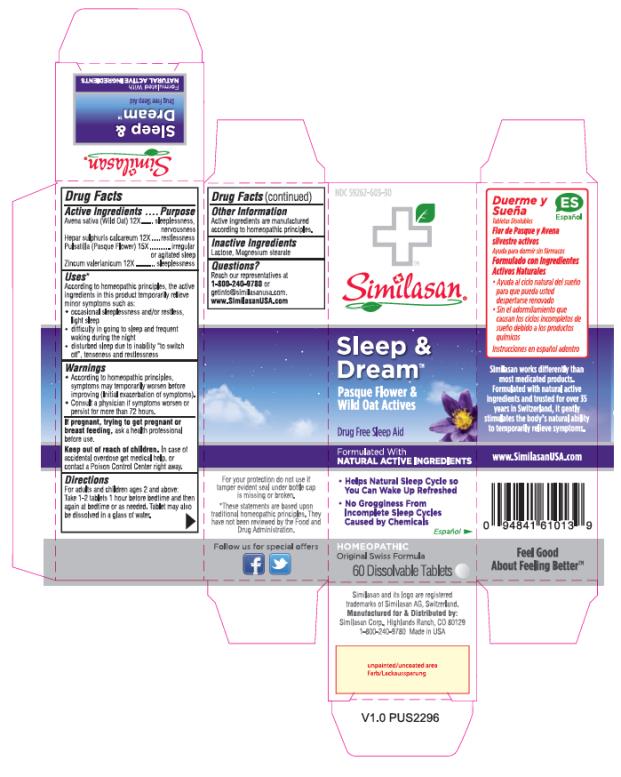
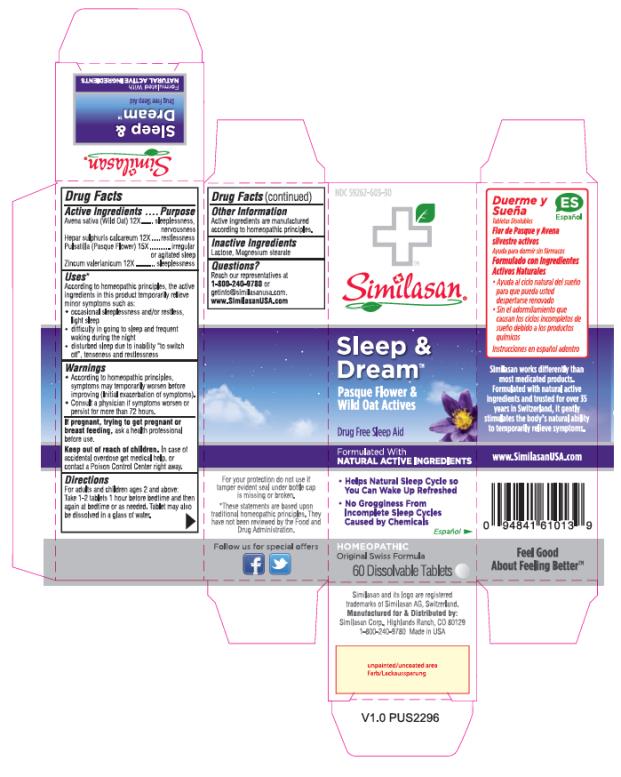
Label: SLEEP AND DREAM- anemone pulsatilla, avena sativa flowering top, calcium sulfide and zinc valerate dihydrate tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 59262-303-30 - Packager: Similasan Corporation
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 12, 2016
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
- Active Ingredients
- Purpose
- Active Ingredients
- Purpose
- Active Ingredients
- Purpose
-
Uses*
According to homeopathic principles, the active ingredients in this product temporarily relieve minor symptoms such as:
• occasional sleeplessness and/or restless, light sleep
• difficulty in going to sleep and frequent waking during the night
• disturbed sleep due to inability “to switch off”, tenseness and restlessness
- Warnings
- Directions
- Other Information
- Inactive Ingredients
- Questions?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SLEEP AND DREAM
anemone pulsatilla, avena sativa flowering top, calcium sulfide and zinc valerate dihydrate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59262-303 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVENA SATIVA FLOWERING TOP (UNII: MA9CQJ3F7F) (AVENA SATIVA FLOWERING TOP - UNII:MA9CQJ3F7F) AVENA SATIVA FLOWERING TOP 12 [hp_X] CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 12 [hp_X] ANEMONE PULSATILLA (UNII: I76KB35JEV) (ANEMONE PULSATILLA - UNII:I76KB35JEV) ANEMONE PULSATILLA 15 [hp_X] ZINC VALERATE DIHYDRATE (UNII: MN0RX54EQA) (VALERIC ACID - UNII:GZK92PJM7B) ZINC VALERATE DIHYDRATE 12 [hp_X] Inactive Ingredients Ingredient Name Strength LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color WHITE Score no score Shape ROUND Size 8mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59262-303-30 60 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED HOMEOPATHIC 02/01/2017 Labeler - Similasan Corporation (111566530)