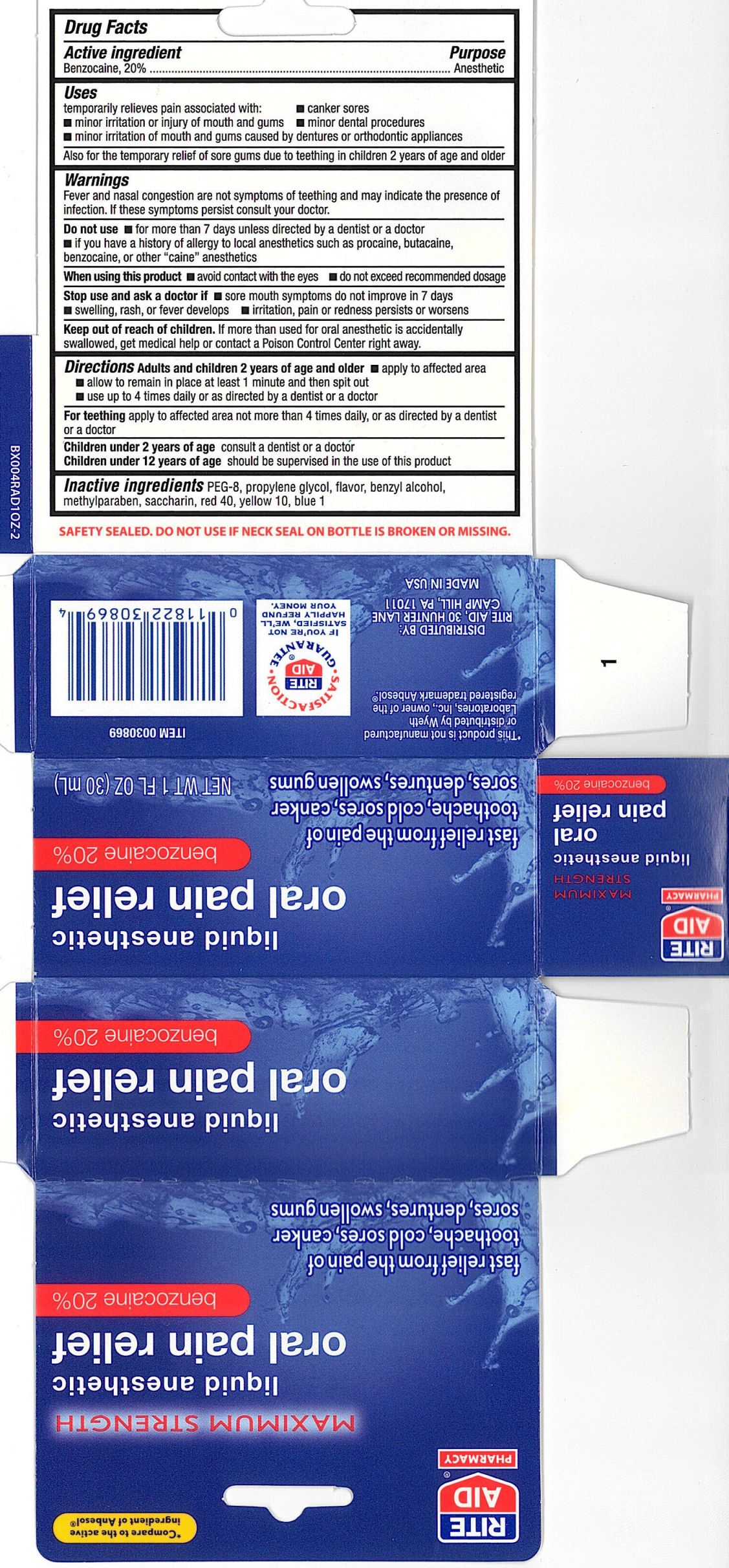
Label: RITE AID LIQUID ANESTHETIC ORAL PAIN RELIEF- benzocaine gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 11822-3950-6 - Packager: Rite Aid Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 8, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
PURPOSE
Uses
- temporary relieves pain associated with: - canker sores
- minor irritation or injury of mouth and gums - minor dental procedures
- minor irritation of mouth and gums caused by dentures or orthodontic appliances
also for the temporary relief of sore gums due to teething in children 2 years of age and older
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Uses
- temporary relieves pain associated with: - canker sores
- minor irritation or injury of mouth and gums - minor dental procedures
- minor irritation of mouth and gums caused by dentures or orthodontic appliances
also for the temporary relief of sore gums due to teething in children 2 years of age and older
-
WARNINGS
Warnings
Fever and nasal congestion are not symptoms of teething and may indicate the presence
of infection. If these symptoms persist, contact your doctor.
Do not use - for more than directed - for more than 7 days, unless directed by a dentist or doctor
- if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or
other "caine" anesthetics
When using this product- avoid contact with the eyes - do not exceed recommended dosage
Stop use and ask a doctor if:
- sore mouth symptoms do not improve in 7 days
- swelling, rash or fever develops - irritation, pain or redness persists or worsens
Keep out of reach of children.If more then used for oral anesthetic is accidentally swallowed,
get medical help or contact a Poison Control Center right away. -
DOSAGE & ADMINISTRATION
Directions Adults and children 2 years of age and older
- apply to the affected area - allow to remain in place at least 1 minute and then spit out
- use up to four times a day or as directed by a dentist or doctor
For teething apply to affected area not more than 4 times dialy, or as directed by a dentist or doctor
Children under 2 years of age consult a dentist or doctor
Children under 12 years of age should be supervised in the use of this product
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RITE AID LIQUID ANESTHETIC ORAL PAIN RELIEF
benzocaine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11822-3950 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C RED NO. 40 (UNII: WZB9127XOA) SPEARMINT OIL (UNII: C3M81465G5) PEPPERMINT OIL (UNII: AV092KU4JH) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SACCHARIN (UNII: FST467XS7D) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11822-3950-6 1 in 1 CARTON 1 30 mL in 1 TUBE, WITH APPLICATOR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part356 07/08/2010 Labeler - Rite Aid Corporation (014578892) Registrant - Pharma Pac, LLC (140807475) Establishment Name Address ID/FEI Business Operations Pharma Pac, LLC 140807475 manufacture