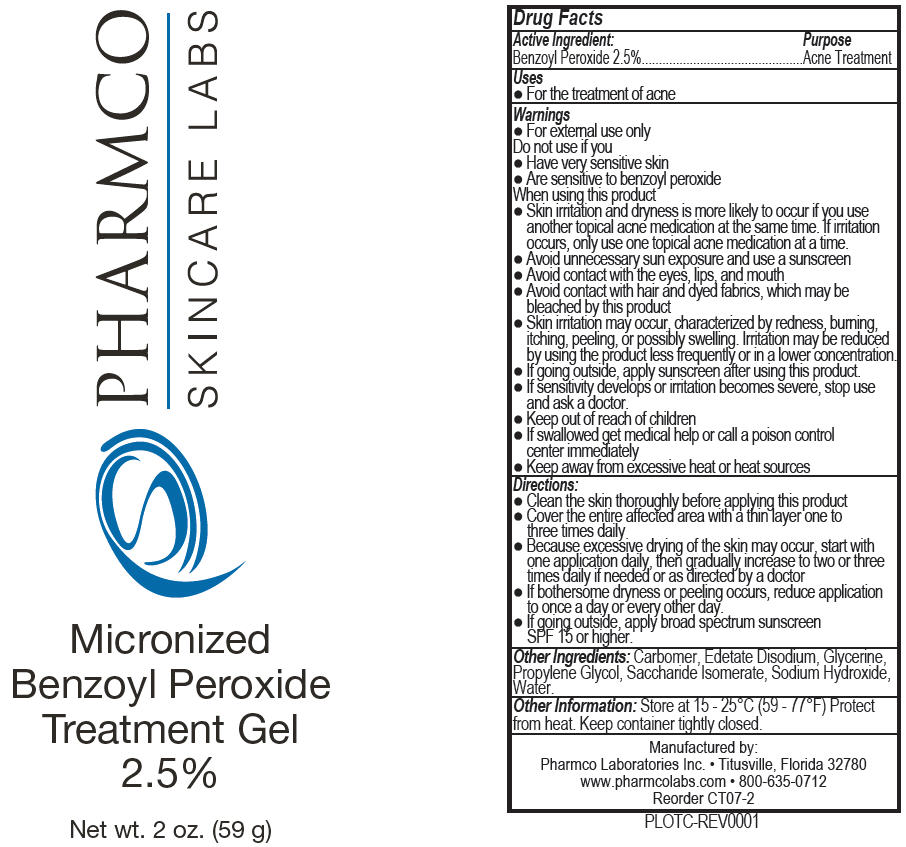
Label: MICRONIZED BENZOYL PEROXIDE TREATMENT- benzoyl peroxide gel
- NDC Code(s): 58400-001-01, 58400-001-02
- Packager: Pharmco Laboratories Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
-
Warnings
- For external use only
When using this product
- Skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- Avoid unnecessary sun exposure and use a sunscreen
- Avoid contact with the eyes, lips, and mouth
- Avoid contact with hair and dyed fabrics, which may be bleached by this product
- Skin irritation may occur, characterized by redness, burning, itching, peeling, or possibly swelling. Irritation may be erduced by using the product less frequently or in a lower concentartion.
- If going outside, apply sunscreen after using this product.
-
Directions
- Clean the skin thoroughly before applying this product
- Cover the entire affected area with a thin layer one to three times daily
- Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- If bothersome dryness or peeling occurs, reduce application to once a day or every other day.
- If going outside, apply broad spectrum sunscreen SPF 15 or higher.
- Other Ingredients
- Other Information
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 59 g Tube Label
-
INGREDIENTS AND APPEARANCE
MICRONIZED BENZOYL PEROXIDE TREATMENT
benzoyl peroxide gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58400-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Benzoyl Peroxide (UNII: W9WZN9A0GM) (Benzoyl Peroxide - UNII:W9WZN9A0GM) Benzoyl Peroxide 25 mg in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Glycerin (UNII: PDC6A3C0OX) Propylene Glycol (UNII: 6DC9Q167V3) Edetate Disodium (UNII: 7FLD91C86K) Sodium Hydroxide (UNII: 55X04QC32I) Saccharide Isomerate (UNII: W8K377W98I) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58400-001-01 59 g in 1 TUBE; Type 0: Not a Combination Product 06/01/2012 2 NDC:58400-001-02 3900 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 06/01/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph drug M006 06/01/2012 Labeler - Pharmco Laboratories Inc. (096270814) Establishment Name Address ID/FEI Business Operations Pharmco Laboratories Inc. 096270814 MANUFACTURE(58400-001) , LABEL(58400-001) , PACK(58400-001) , ANALYSIS(58400-001)