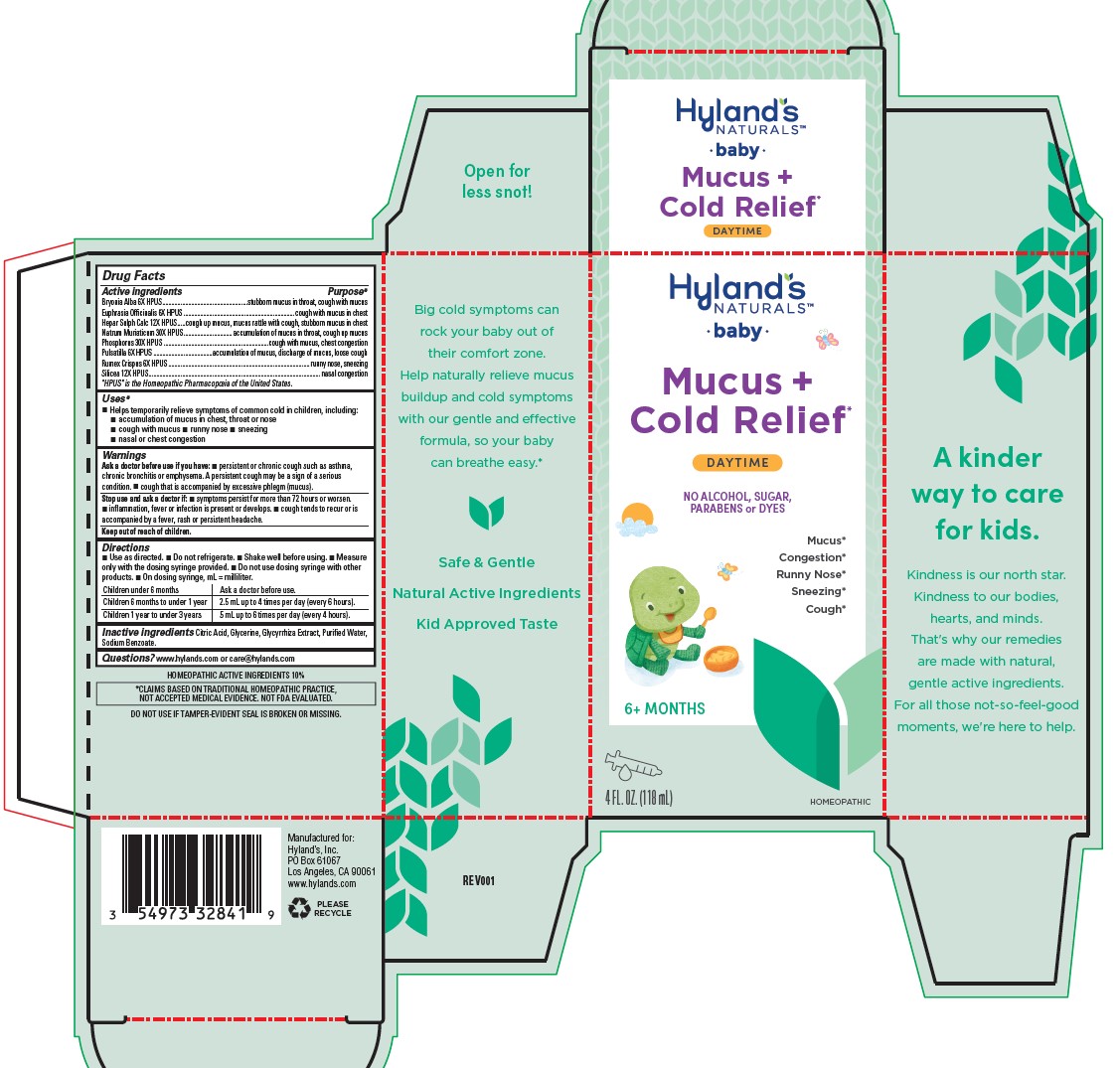
Label: BABY MUCUS PLUS COLD RELIEF- bryonia alba root, euphrasia stricta, calcium sulfide, sodium chloride, phosphorus, anemone pulsatilla, rumex crispus root, and silicon dioxide liquid
- NDC Code(s): 54973-3284-1, 54973-3284-2, 54973-3284-3
- Packager: Hyland's Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated August 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Drug Facts
Active ingredients Purpose* Bryonia Alba 6X HPUS stubborn mucus in throat, cough with mucus Euphrasia Officinalis 6X HPUS cough with mucus in chest Hepar Sulph Calc 12X HPUS cough up mucus, mucus rattle with cough, stubborn mucus in chest Natrum Muriaticum 30X HPUS accumulation of mucus in throat, cough up mucus Phosphorus 30X HPUS cough with mucus, chest congestion Pulsatilla 6X HPUS accumulation of mucus, discharge of mucus, loose cough Rumex Crispus 6X HPUS runny nose, sneezing Silicea 12X HPUS nasal congestion "HPUS" is the Homeopathic Pharmacopoeia of the United States.
- Uses*
-
Warnings
Ask a doctor before use if you have
■ persistent or chronic cough such as asthma, chronic bronchitis or emphysema. A persistent cough may be a sign of a serious
condition.■ cough that is accompanied by excessive phlegm (mucus).
-
Directions
■ Use as directed. ■ Do not refrigerate. ■ Shake well before using. Measure
only with the dosing syringe provided. ■ Do not use dosing syringe with other
products. ■ On dosing syringe, mL = milliliter.Children under 6 months Ask a doctor before use. Children 6 months to under 1 year 2.5 mL up to 4 times per day (every 6 hours). Children 1 year to under 3 years 5 mL up to 6 times per day (every 4 hours). - Inactive ingredients
- Questions?
- HOMEOPATHIC ACTIVE INGREDIENTS 10%
- SPL UNCLASSIFIED SECTION
- DO NOT USE IF TAMPER-EVIDENT SEAL IS BROKEN OR MISSING.
- PURPOSE
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
BABY MUCUS PLUS COLD RELIEF
bryonia alba root, euphrasia stricta, calcium sulfide, sodium chloride, phosphorus, anemone pulsatilla, rumex crispus root, and silicon dioxide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54973-3284 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 6 [hp_X] in 1 mL EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 6 [hp_X] in 1 mL CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 12 [hp_X] in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 30 [hp_X] in 1 mL PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 30 [hp_X] in 1 mL ANEMONE PULSATILLA (UNII: I76KB35JEV) (ANEMONE PULSATILLA - UNII:I76KB35JEV) ANEMONE PULSATILLA 6 [hp_X] in 1 mL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 12 [hp_X] in 1 mL RUMEX CRISPUS ROOT (UNII: 9N1RM2S62C) (RUMEX CRISPUS ROOT - UNII:9N1RM2S62C) RUMEX CRISPUS ROOT 6 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) GLYCERIN (UNII: PDC6A3C0OX) LICORICE (UNII: 61ZBX54883) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54973-3284-1 1 in 1 CARTON 05/13/2016 1 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:54973-3284-2 2.5 mL in 1 POUCH; Type 0: Not a Combination Product 12/23/2016 12/20/2021 3 NDC:54973-3284-3 1 in 1 CARTON 08/01/2018 3 148 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/13/2016 Labeler - Hyland's Inc. (008316655) Establishment Name Address ID/FEI Business Operations Hyland's Inc. 008316655 manufacture(54973-3284) , pack(54973-3284) Establishment Name Address ID/FEI Business Operations Applied Laboratories, Inc. 117337220 pack(54973-3284)