Label: OPITOX TOPICAL GEL- menthol camphor gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 72380-521-10 - Packager: Vivera Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 5, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- OTHER SAFETY INFORMATION
- ASK DOCTOR
-
WHEN USING
When using this product: Avoid contact with the
eyes and/or mucous membranes; Not for ophthalmic
use; Do not apply to wounds or damaged skin; Do not
use with other sprays, ointments, creams or liniments;
Do not apply to irritated skin or if excessive irritation
develops; Do not bandage; Do not use with heating
pads or device. - STOP USE
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive Ingredients: SD Alcohol 40-B, Water,
Carbomer, Glycerin, Cannabis Sativa (Hemp) Seed Oil,
Cannabidiol (CBD), Aloe Barbadensis Leaf Juice, Arnica
Montana Flower Extract, Mentha Piperita (Peppermint)
Oil, Mentha Viridis (Spearmint) Leaf Oil, Propylene
Glycol, Hamamelis Virginiana (Witch Hazel) Extract,
Melaleuca Alternifolia (Tea Tree) Leaf Oil, Capsicum
Annuum Fruit Extract, Camellia Sinensis (Green
Tea) Leaf Extract, Phenoxyethanol, Caprylyl Glycol,
Ethylhexylglycerin, Hexylene Glycol, Triethanolamine - QUESTIONS
-
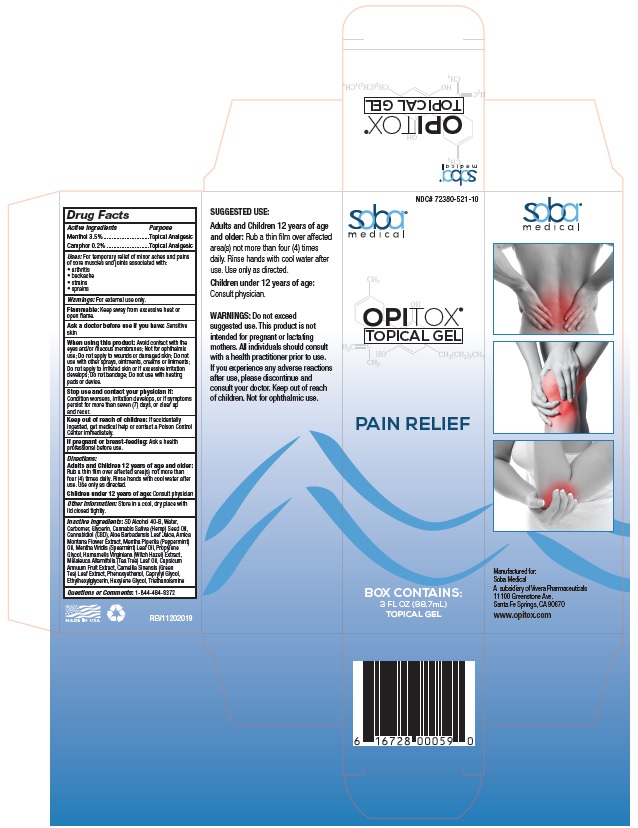
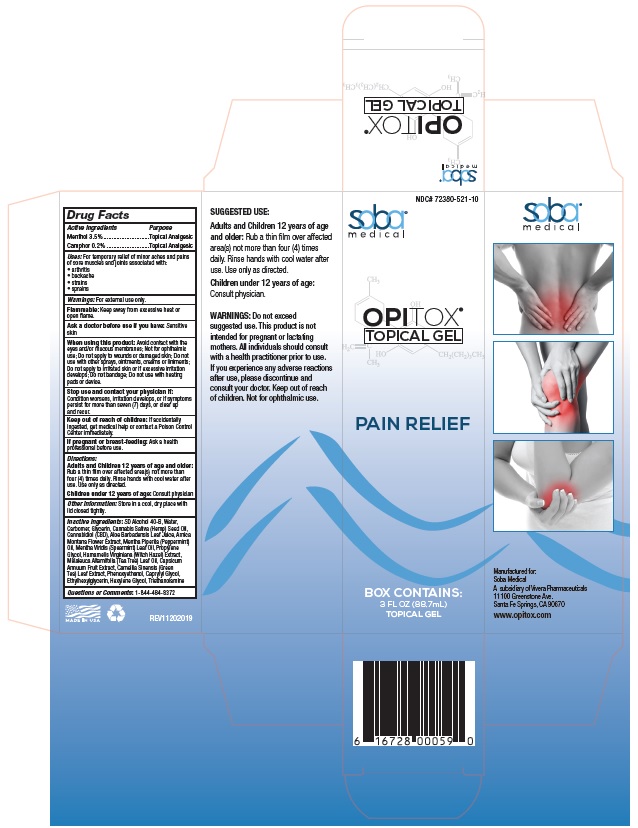
Principal Display Package
NDC# 72380-521-10
soba®
medicalOPITOX®
TOPICAL GELPAIN RELIEF
BOX CONTAINS:
3 FL OZ (88.7mL)
TOPICAL GEL6 16728 00059 0
SUGGESTED USE:
Adults and Children 12 years of age
and older: Rub a thin film over affected
area(s) not more than four (4) times
daily. Rinse hands with cool water after
use. Use only as directed.
Children under 12 years of age:
Consult physician.WARNINGS: Do not exceed
suggested use. This product is not
intended for pregnant or lactating
mothers. All individuals should consult
with a health practitioner prior to use.
If you experience any adverse reactions
after use, please discontinue and
consult your doctor. Keep out of reach
of children. Not for ophthalmic use.soba®
medicalManufactured for:
Soba Medical
A subsidiary of Vivera Pharmaceuticals
11100 Greenstone Ave. Santa Fe Springs, CA 90670
www.opitox.comMADE IN USA REV11202019
soba®
medicalOPITOX®
TOPICAL GELRetail Box
Tube

Information Insert

res
-
INGREDIENTS AND APPEARANCE
OPITOX TOPICAL GEL
menthol camphor gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72380-521 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 3.5 g in 100 mL CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 0.2 g in 100 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) CANNABIS SATIVA SEED OIL (UNII: 69VJ1LPN1S) CANNABIDIOL (UNII: 19GBJ60SN5) ALOE VERA LEAF (UNII: ZY81Z83H0X) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) PEPPERMINT OIL (UNII: AV092KU4JH) SPEARMINT OIL (UNII: C3M81465G5) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) HAMAMELIS VIRGINIANA LEAF (UNII: T07U1161SV) TEA TREE OIL (UNII: VIF565UC2G) PAPRIKA (UNII: X72Z47861V) GREEN TEA LEAF (UNII: W2ZU1RY8B0) PHENOXYETHANOL (UNII: HIE492ZZ3T) CAPRYLIC ALCOHOL (UNII: NV1779205D) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) HEXYLENE GLYCOL (UNII: KEH0A3F75J) TROLAMINE (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72380-521-10 1 in 1 BOX 08/06/2019 1 88.7 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 08/06/2019 Labeler - Vivera Pharmaceuticals, Inc. (081244342)