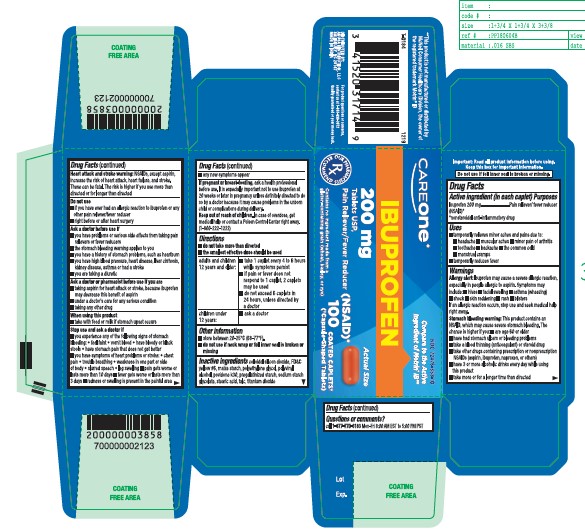
Label: IBUPROFEN tablet
- NDC Code(s): 72476-358-05, 72476-358-10
- Packager: RETAIL BUSINESS SERVICES,LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Allergy alert:
-
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
■ are age 60 or older
■ have had stomach ulcers or bleeding problems
■ take a blood thinning (anticoagulant) or steroid drug
■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
■ have 3 or more alcoholic drinks every day while using this product
■ take more or for a longer time than directed
- Heart attack and stroke warning:
- Do not use
-
Ask a doctor before use if
■ you have problems or serious side effects from taking pain relievers or fever reducers
■ the stomach bleeding warning applies to you
■ you have a history of stomach problems, such as heartburn
■ you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, or had a stroke
■ you are taking a diuretic
- Ask a doctor or pharmacist before use if you are
- When using this product
-
Stop use and ask a doctor if
■ you experience any of the following signs of stomach bleeding:
■ feel faint
■ vomit blood
■ have bloody or black stools
■ have stomach pain that does not get better
■ you have symptoms of heart problems or stroke:
■ chest pain
■ trouble breathing
■ weakness in one part or side of body
■ slurred speech
■ leg swelling
■ pain gets worse or lasts more than 10 days
■ fever gets worse or lasts more than 3 days
■ redness or swelling is present in the painful area
■ any new symptoms appear
- If pregnant or breast-feeding,
- Keep out of the reach of children.
-
Directions
■ do not take more than directed
■ the smallest effective dose should be used
Adults and children 12 years and older
■ take 1 caplet every 4 to 6 hours while symptoms persist
■ if pain or fever does not respond to 1 caplet, 2 caplets may be used
■ do not exceed 6 caplets in 24 hours, unless directed by a doctor
Children under 12 years
■ ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
- PDP
-
INGREDIENTS AND APPEARANCE
IBUPROFEN
ibuprofen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72476-358 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) STARCH, CORN (UNII: O8232NY3SJ) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STEARIC ACID (UNII: 4ELV7Z65AP) POVIDONE K30 (UNII: U725QWY32X) Product Characteristics Color orange Score no score Shape OVAL (Caplet shaped film coated tablets) Size 14mm Flavor Imprint Code G;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72476-358-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 07/26/2020 2 NDC:72476-358-05 50 in 1 BOTTLE; Type 0: Not a Combination Product 07/26/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202312 07/26/2020 Labeler - RETAIL BUSINESS SERVICES,LLC. (967989935)