Label: RESTORAL- menthol ointment
- NDC Code(s): 69204-023-01, 69204-023-02
- Packager: Natures Formulae Health Products Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 16, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
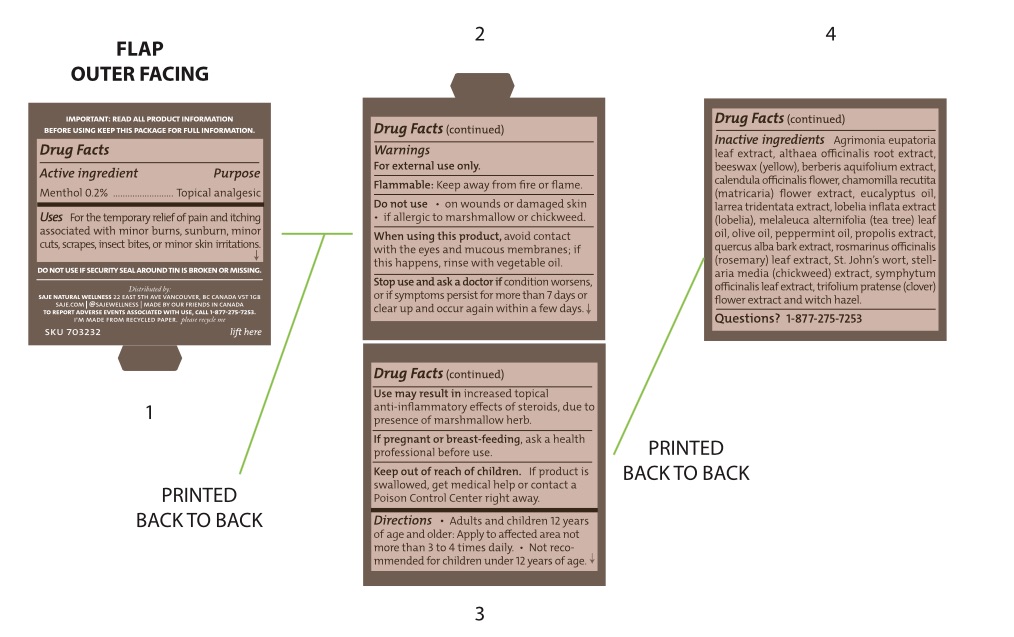
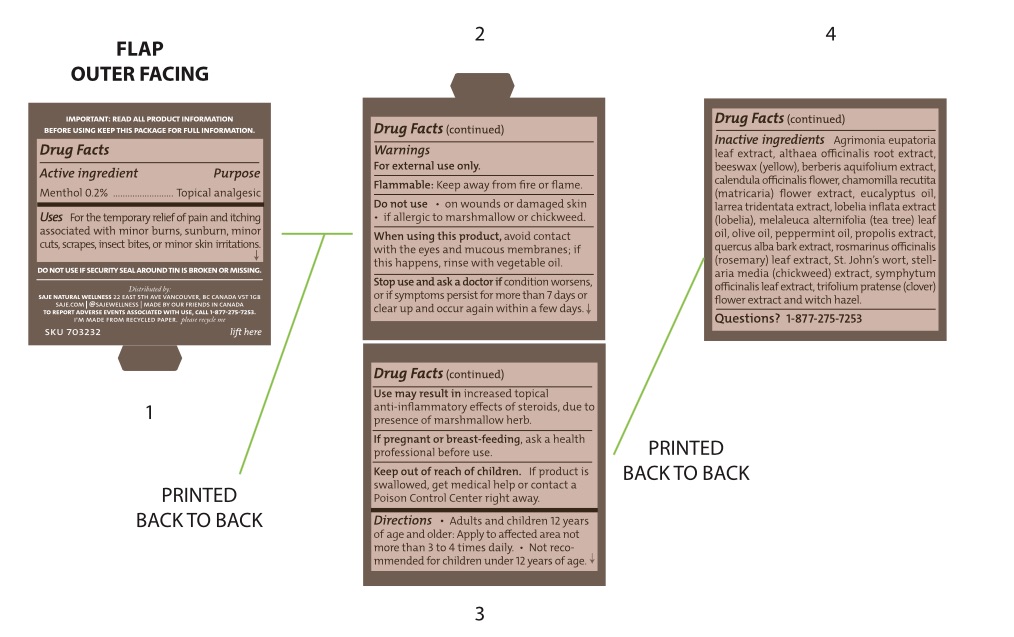
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Flammable - keep away from fire or flame
Use may result in increased topical anti-inflammatory effects of steroids, due to presence of marshmallow herb
When using this product
Avoid contact with eyes and mucous membranes; if this happens, rinse with vegetable oil
- Directions
-
Inactive ingredients
Agrimonia eupatoria leaf extract, althaea officinalis root extract, beeswax (yellow), berberis aquifolium extract, calendula officinalis flower, chamomilla recutita (matricaria) flower extract, eucalyptus oil, larrea tridentata extract, lobelia inflata extract (lobelia), melaleuca alternifolia (tea tree) leaf oil, olive oil, peppermint oil, propolis extract, quercus alba bark extract, rosmarinus officinalis (rosemary) leaf extract, St. John’s wort, stellaria media (chickweed) extract, symphytum officinalis leaf extract, trifolium pratense (clover) flower extract and witch hazel
- Questions?
- Principle Display Panel information
- Restoral Topical analgesic ointment - Outer label
-
INGREDIENTS AND APPEARANCE
RESTORAL
menthol ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69204-023 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.2 g in 100 g Inactive Ingredients Ingredient Name Strength PROPOLIS WAX (UNII: 6Y8XYV2NOF) OLIVE OIL (UNII: 6UYK2W1W1E) COMFREY ROOT (UNII: M9VVZ08EKQ) PEPPERMINT OIL (UNII: AV092KU4JH) YELLOW WAX (UNII: 2ZA36H0S2V) ST. JOHN'S WORT (UNII: UFH8805FKA) STELLARIA MEDIA (UNII: 2H03479QVR) TRIFOLIUM PRATENSE FLOWER (UNII: 4JS0838828) ALTHAEA OFFICINALIS ROOT (UNII: TRW2FUF47H) LARREA TRIDENTATA LEAF (UNII: PK0TXD049P) BERBERIS AQUIFOLIUM ROOT (UNII: 746TB9VNDP) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) EUCALYPTUS OIL (UNII: 2R04ONI662) TEA TREE OIL (UNII: VIF565UC2G) AGRIMONIA EUPATORIA LEAF (UNII: 8Z160YZ4AP) QUERCUS ALBA BARK (UNII: 93LP7Y8EGT) ROSEMARY (UNII: IJ67X351P9) WITCH HAZEL (UNII: 101I4J0U34) MATRICARIA CHAMOMILLA FLOWERING TOP (UNII: 3VNC7T6Z02) LOBELIA INFLATA LEAF (UNII: 7QFT17RLRG) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69204-023-01 1 in 1 BOX 09/12/2018 1 54.1 g in 1 CONTAINER; Type 0: Not a Combination Product 2 NDC:69204-023-02 13.5 g in 1 CONTAINER; Type 0: Not a Combination Product 09/12/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 09/12/2018 Labeler - Natures Formulae Health Products Ltd. (241385587) Establishment Name Address ID/FEI Business Operations Natures Formulae Health Products Ltd. 241385587 manufacture(69204-023)