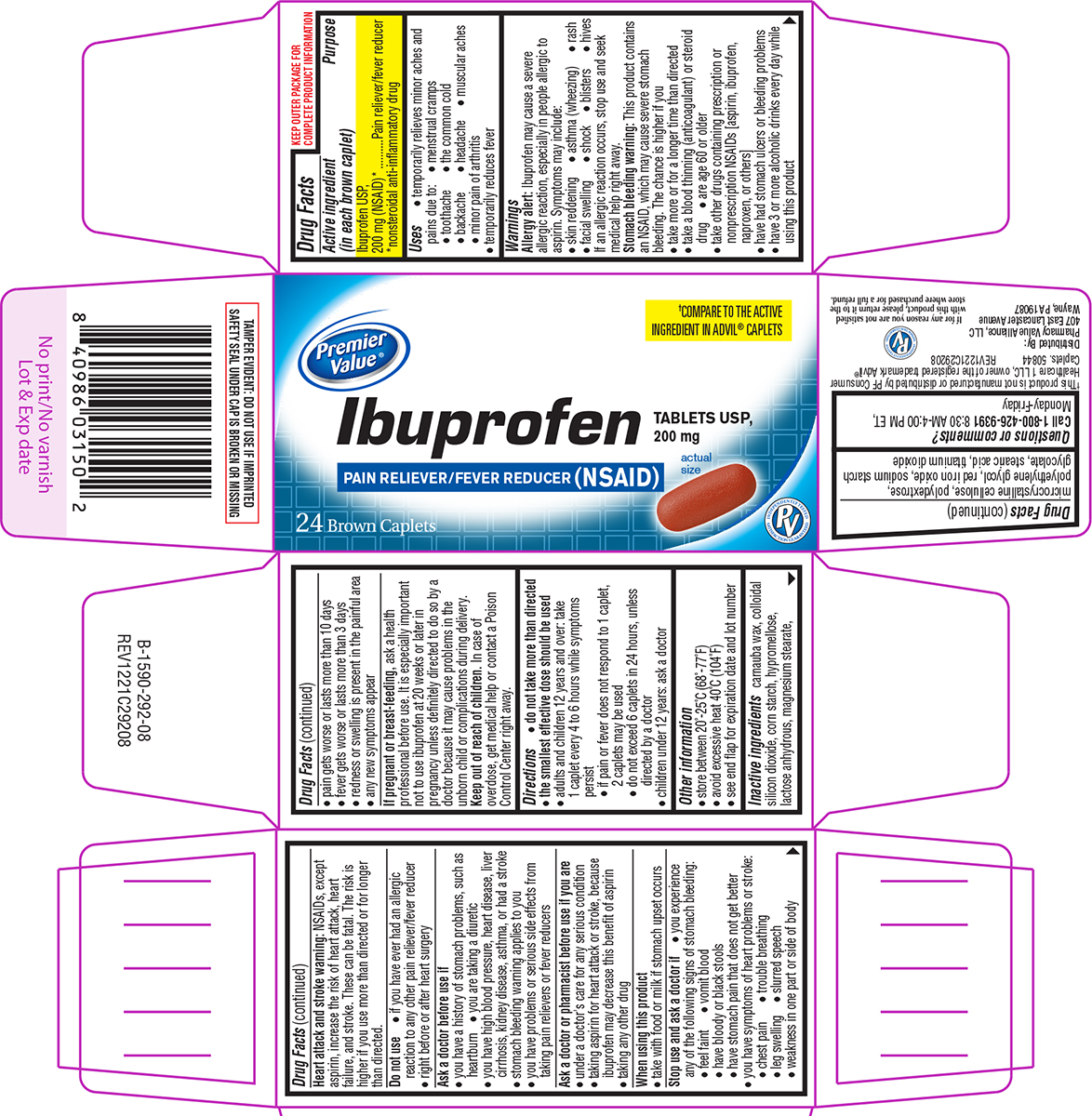
Label: IBUPROFEN tablet, film coated
- NDC Code(s): 68016-645-08, 68016-645-12, 68016-645-15
- Packager: Chain Drug Consortium
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each brown caplet)
- Purpose
- Uses
-
Warnings
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- skin reddening
- asthma (wheezing)
- rash
- facial swelling
- shock
- blisters
- hives
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- take more or for a longer time than directed
- take a blood thinning (anticoagulant) or steroid drug
- are age 60 or older
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have had stomach ulcers or bleeding problems
- have 3 or more alcoholic drinks every day while using this product
Heart attack and stroke warning: NSAIDs, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be fatal. The risk is higher if you use more than directed or for longer than directed.
Do not use
- right before or after heart surgery
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
Ask a doctor before use if
- you have a history of stomach problems, such as heartburn
- you are taking a diuretic
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, or had a stroke
- stomach bleeding warning applies to you
- you have problems or serious side effects from taking pain relievers or fever reducers
Ask a doctor or pharmacist before use if you are
- under a doctor's care for any serious condition
- taking aspirin for heart attack or stroke, because ibuprofen may decrease this benefit of aspirin
- taking any other drug
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- you have symptoms of heart problems or stroke
- chest pain
- trouble breathing
- leg swelling
- slurred speech
- weakness in one part or side of body
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present in the painful area
- any new symptoms appear
-
Directions
- do not take more than directed
- the smallest effective dose should be used
- adults and children 12 years and over: take 1 caplet every 4 to 6 hours while symptoms persist
- if pain or fever does not respond to 1 caplet, 2 caplets may be used
- do not exceed 6 caplets in 24 hours, unless directed by a doctor
- children under 12 years: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
Premier
Value®†COMPARE TO THE ACTIVE
INGREDIENT IN ADVIL® CAPLETSIbuprofen
TABLETS USP,
200 mg
PAIN RELIEVER/FEVER REDUCER (NSAID)24 Brown Caplets
actual
sizeTAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING†This product is not manufactured or distributed by PF Consumer
Healthcare 1 LLC, owner of the registered trademark Advil®
Caplets. 50844 REV1221C29208Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue
Wayne, PA 19087If for any reason you are not satisfied
with this product, please return it to the
store where purchased for a full refund.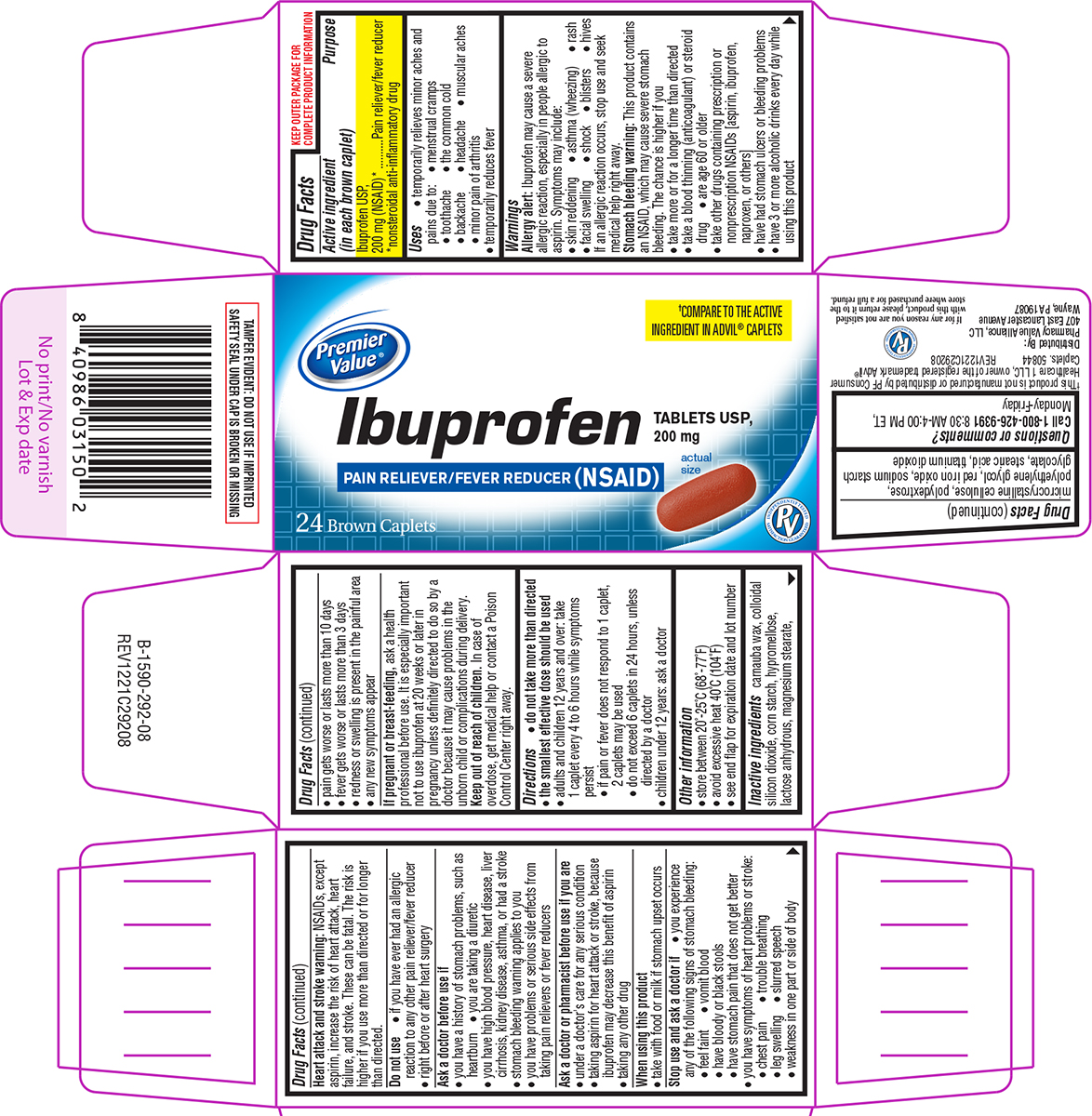
Premier Value 44-292
-
INGREDIENTS AND APPEARANCE
IBUPROFEN
ibuprofen tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-645 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) FERRIC OXIDE RED (UNII: 1K09F3G675) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color brown Score no score Shape OVAL Size 14mm Flavor Imprint Code 44;292 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-645-08 1 in 1 CARTON 05/24/1988 1 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:68016-645-15 1 in 1 CARTON 05/24/1988 2 50 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:68016-645-12 1 in 1 CARTON 05/24/1988 3 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075010 05/24/1988 Labeler - Chain Drug Consortium (101668460) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(68016-645) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(68016-645) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(68016-645) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(68016-645) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(68016-645)