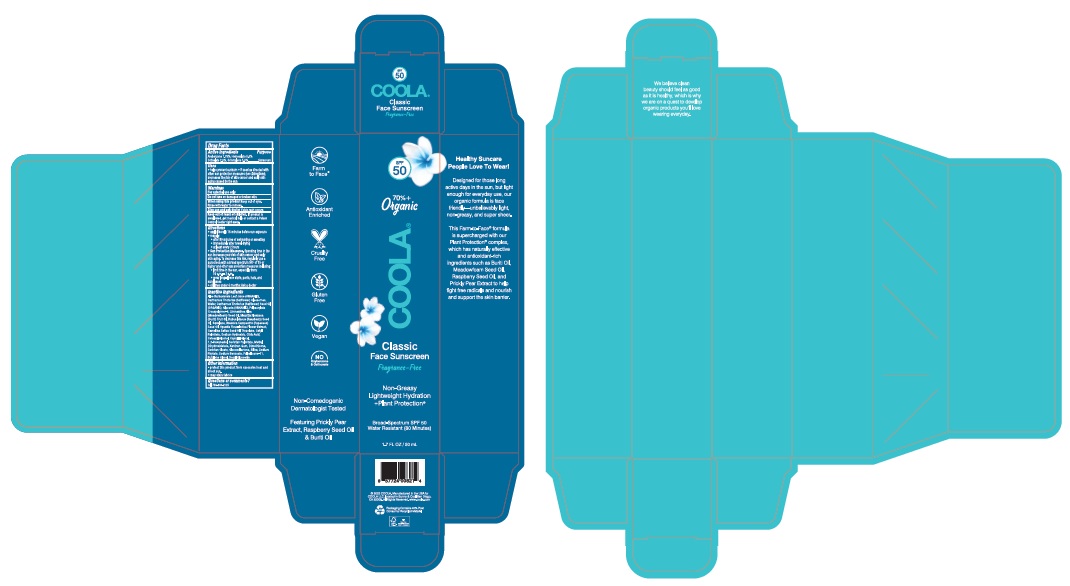
Label: COOLA CLASSIC FACE SUNSCREEN FRAGRANCE-FREE SPF 50- avobenzone, homosalate, octisalate, octocrylene lotion
- NDC Code(s): 79753-074-01
- Packager: COOLA LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS AND PRECAUTIONS
-
DOSAGE & ADMINISTRATION
Directions
* apply liberally 15 minutes before sun exposure
* reapply:
* after 80 minutes of swimming or sweating
* immediately after towel drying
* at least every 2 hours
* Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
* limit time in the sun, especially from: 10 a.m. – 2 p.m.
* wear long-sleve shirts, pants, hats, and sunglasses* children under 6 months: Ask a doctor
-
INACTIVE INGREDIENT
Inactive ingredients
Aloe Barbadensis Leaf Juice (ORGANIC),
Carthamus Tinctorius (Safflower) Oleosomes,
Water, Carthamus Tinctorius (Safflower) Seed Oil
(ORGANIC), Glycerin (ORGANIC), Polyacrylate
Crosspolymer-6, Limnanthes Alba
(Meadowfoam) Seed Oil, Mauritia Flexuosa
(Buriti) Fruit Oil, Rubus Idaeus (Raspberry) Seed
Oil, Squalane, Brassica Campestris (Rapeseed)
Seed Oil, Opuntia Ficus-Indica Flower Extract,
Camelina Sativa Seed Oil, Tropolone, Cetyl
Palmitate, Sodium Hydroxide, Citric Acid,
Cetearyl Alcohol, Caprylyl Glycol,
1,2-Hexanediol, Sorbitan Palmitate, Methyl
Dihydroabietate, Xanthan Gum, Dimethicone,
Sorbitan Oleate, Gluconolactone, Silica, Sodium
Phytate, Sodium Benzoate, Polysilicone-11,
Butylene Glycol, Decyl Glucoside - OTHER SAFETY INFORMATION
- QUESTIONS
-
Product Labeling - COOLA Classic Face Sunscreen Fragrance-Free SPF 50
SPF
5070% +
OrganicCOOLA
Classic
Face Sunscreen
FragranceiFreeBroad Spectrum SPF 50
Water Resistant (80 Minutes)1.7 FL OZ / 50 mL
2022 COOLA. Manufactured in the USA forDistributed by: COOLA LLC,
COOLA LLC, located in Sunny& Cool San Diego,
CA 92009. All Rights Reserved. ww.coola.com
Carton
Tube

-
INGREDIENTS AND APPEARANCE
COOLA CLASSIC FACE SUNSCREEN FRAGRANCE-FREE SPF 50
avobenzone, homosalate, octisalate, octocrylene lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79753-074 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 20 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 50 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 50 mg in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 17.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) XANTHAN GUM (UNII: TTV12P4NEE) WATER (UNII: 059QF0KO0R) TROPOLONE (UNII: 7L6DL16P1T) PHYTATE SODIUM (UNII: 88496G1ERL) ALOE VERA LEAF (UNII: ZY81Z83H0X) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) SQUALANE (UNII: GW89575KF9) BRASSICA RAPA SUBSP. OLEIFERA OIL (UNII: N4G8379626) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) SORBITAN MONOPALMITATE (UNII: 77K6Z421KU) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) METHYL DIHYDROABIETATE (UNII: 7666FJ0J9F) SODIUM BENZOATE (UNII: OJ245FE5EU) RASPBERRY SEED OIL (UNII: 9S8867952A) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) AMMONIUM ACRYLOYLDIMETHYLTAURATE, DIMETHYLACRYLAMIDE, LAURYL METHACRYLATE AND LAURETH-4 METHACRYLATE COPOLYMER, TRIMETHYLOLPROPANE TRIACRYLATE CROSSLINKED (45000 MPA.S) (UNII: Q7UI015FF9) OPUNTIA FICUS-INDICA (UNII: 23Z87HTQ6P) GLUCONOLACTONE (UNII: WQ29KQ9POT) CARTHAMUS TINCTORIUS SEEDCAKE (UNII: DHQ13F4N2W) DIMETHICONE (UNII: 92RU3N3Y1O) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) GLYCERIN (UNII: PDC6A3C0OX) SAFFLOWER OIL (UNII: 65UEH262IS) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CAMELINA SATIVA SEED OIL (UNII: 12824X01L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79753-074-01 1 in 1 CARTON 02/01/2023 1 50 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 02/01/2023 Labeler - COOLA LLC (956990290) Registrant - Bentley Laboratories, LLC (068351753)