Label: CANDIDA- ammoniac, aspergillus niger var. niger, caffeic acid, candida albicans, candida parapsilosis, ipecac, lycopodium clavatum spore, yeast mannan, vaccinium vitis-idaea leaf spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 63776-073-15 - Packager: VIATREXX BIO INCORPORATED
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated November 30, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose:
- Uses
- Warnings
- Dosage
- Other Ingredients
- Other Information
- Questions
-
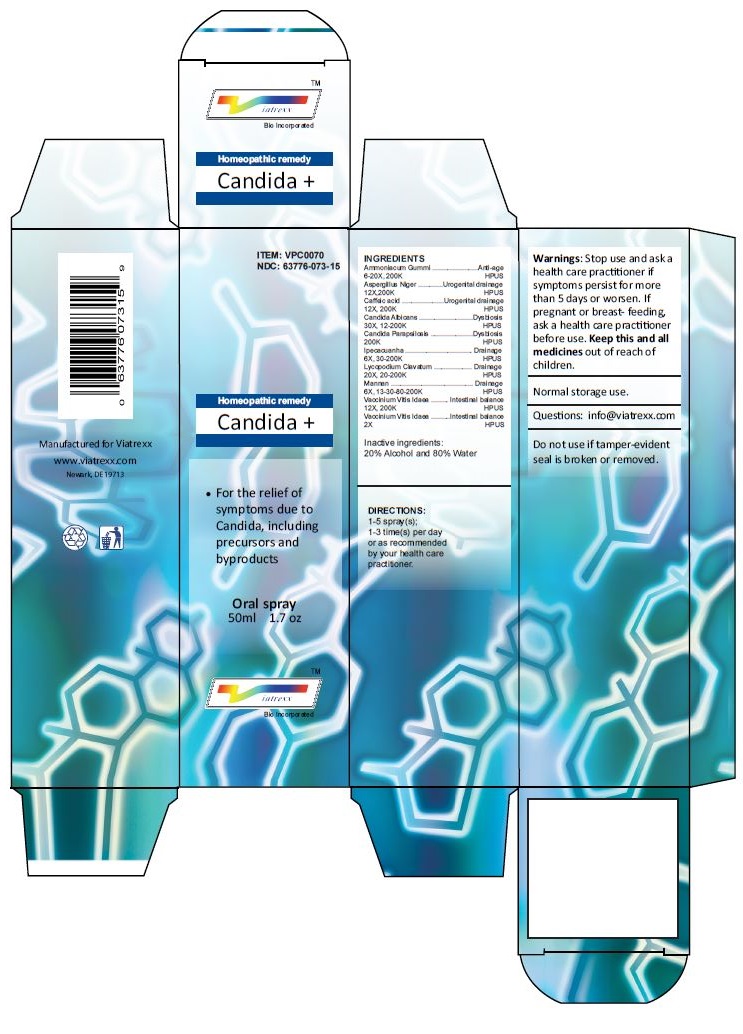
Principal Display Panel
ITEM: VPC0070
NDC 63776-073-15
Homeopathic remedy
Candida +
• For the relief of symptoms due to Candida, including precursors and byproducts
Oral spray
50ml 1.7 oz
Viatrexx™ Bio Incorporated
Manufactured by Viatrexx
www.viatrexx.com
Newark, DE 19713
Candida +
50 mL
1.7 oz
Viatrexx™ Bio Incorporated
ITEM: VPC0070
NDC: 63776-073-15
INDICATIONS:
For the relief of symptoms due to Candida, including precursors and byproducts
DIRECTIONS:
1-5 spray(s); 1-3 time(s) per day or as recommended by your health care practitioner.
Mfg. for
Viatrexx Bio Incorporated.
www.viatrexx.com
Newark, DE 19713
-
INGREDIENTS AND APPEARANCE
CANDIDA
ammoniac, aspergillus niger var. niger, caffeic acid, candida albicans, candida parapsilosis, ipecac, lycopodium clavatum spore, yeast mannan, vaccinium vitis-idaea leaf sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63776-073 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AMMONIAC (UNII: 8471AK50P8) (AMMONIAC - UNII:8471AK50P8) AMMONIAC 200 [kp_C] in 1 mL ASPERGILLUS NIGER VAR. NIGER (UNII: 9IOA40ANG6) (ASPERGILLUS NIGER VAR. NIGER - UNII:9IOA40ANG6) ASPERGILLUS NIGER VAR. NIGER 200 [kp_C] in 1 mL CAFFEIC ACID (UNII: U2S3A33KVM) (CAFFEIC ACID - UNII:U2S3A33KVM) CAFFEIC ACID 200 [kp_C] in 1 mL CANDIDA ALBICANS (UNII: 4D7G21HDBC) (CANDIDA ALBICANS - UNII:4D7G21HDBC) CANDIDA ALBICANS 200 [kp_C] in 1 mL CANDIDA PARAPSILOSIS (UNII: 0KZ676D44N) (CANDIDA PARAPSILOSIS - UNII:0KZ676D44N) CANDIDA PARAPSILOSIS 200 [kp_C] in 1 mL IPECAC (UNII: 62I3C8233L) (IPECAC - UNII:62I3C8233L) IPECAC 200 [kp_C] in 1 mL LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 200 [kp_C] in 1 mL YEAST MANNAN (UNII: 91R887N59P) (YEAST MANNAN - UNII:91R887N59P) YEAST MANNAN 200 [kp_C] in 1 mL VACCINIUM VITIS-IDAEA LEAF (UNII: FO2ACM0RMQ) (VACCINIUM VITIS-IDAEA LEAF - UNII:FO2ACM0RMQ) VACCINIUM VITIS-IDAEA LEAF 200 [kp_C] in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63776-073-15 1 in 1 BOX 07/24/2012 1 50 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 07/24/2012 Labeler - VIATREXX BIO INCORPORATED (078419880) Establishment Name Address ID/FEI Business Operations Les Importations Herbasante Inc 243254612 manufacture(63776-073)