Label: UREA 39.5%- urea cream
- NDC Code(s): 59088-698-16
- Packager: PureTek Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 20, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Urea 39.5% Cream is a keratolytic emollient which is a gentle, yet potent, tissue softener for nails and/or skin. Each gram contains 395 mg of urea in a vehicle consisting of: Aqua (Purified Water), Ceteareth-6, Ceteareth-25, Cetyl Alcohol, Methylparaben, Paraffin, Propylene Glycol, Stearyl Alcohol, Xanthan Gum.
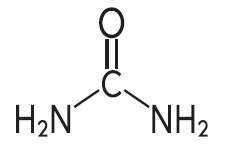
Urea is a diamide of carbonic acid with the following chemical structure:
- CLINICAL PHARMACOLOGY
-
INDICATIONS & USAGE
For debridement and promotion of normal healing of hyperkeratotic surface lesions, particularly where healing is retarded by local infection, necrotic tissue, fibrinous or purulent debris or eshar. Urea is useful for the treatment of hyperkeratotic conditions such as dry, rough skin, dermatitis, psoriasis, xerosis, ichthyosis, eczema, keratosis pilaris, keratosis palmaris, keratoderma, corns and calluses, as well as damaged, ingrown and devitalized nails.
- CONTRAINDICATIONS
-
WARNINGS
FOREXTERNALUSEONLY.NOTFOR OPHTHALMIC USE. KEEP OUT OF REACH OF CHILDREN.
Avoid contact with eyes, lips and mucous membranes.
General
This product is to be used as directed by a physician and should not be used to treat any condition other than that for which it was prescribed. If redness or irritation occurs, discontinue use and consult a physician.
Information for Patients
Patients should discontinue the use of this product if the condition becomes worse or if a rash develops in the area being treated or elsewhere. Avoid contact with eyes, lips and mucous membranes.
Carcinogenesis, Mutagenesis and Impairment of Fertility:
Long-term animal studies for carcinogenic potential have not been performed on this product to date. Studies on reproduction and fertility also have not been performed.
Pregnancy:
Category C. Animal reproduction studies have not been conducted with this product. It is also not known whether this product can affect reproduction capacity or cause fetal harm when administered to a pregnant woman. This product should be used by a pregnant woman only if clearly needed or when potential benefits outweigh potential hazards to the fetus.
- ADVERSE REACTIONS:
- DOSAGE AND ADMINISTRATION
- HOW SUPPLIED
- STORAGE
- Urea 39.5% Cream™
-
INGREDIENTS AND APPEARANCE
UREA 39.5%
urea creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59088-698 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength UREA (UNII: 8W8T17847W) (UREA - UNII:8W8T17847W) UREA 395 mg in 1 g Inactive Ingredients Ingredient Name Strength CETEARETH-25 (UNII: 8FA93U5T67) CETEARETH-6 (UNII: 2RJS3559D3) CETYL ALCOHOL (UNII: 936JST6JCN) METHYLPARABEN (UNII: A2I8C7HI9T) PARAFFIN (UNII: I9O0E3H2ZE) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59088-698-16 227 g in 1 JAR; Type 0: Not a Combination Product 01/20/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/20/2023 Labeler - PureTek Corporation (785961046)


