Label: LONG LAST MAKE UP 02 (NEO HELIOPAN)- ethylhexyl methoxycinnamate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 24653-805-01, 24653-805-02 - Packager: Janssen Cosmetics GmbH
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 7, 2011
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
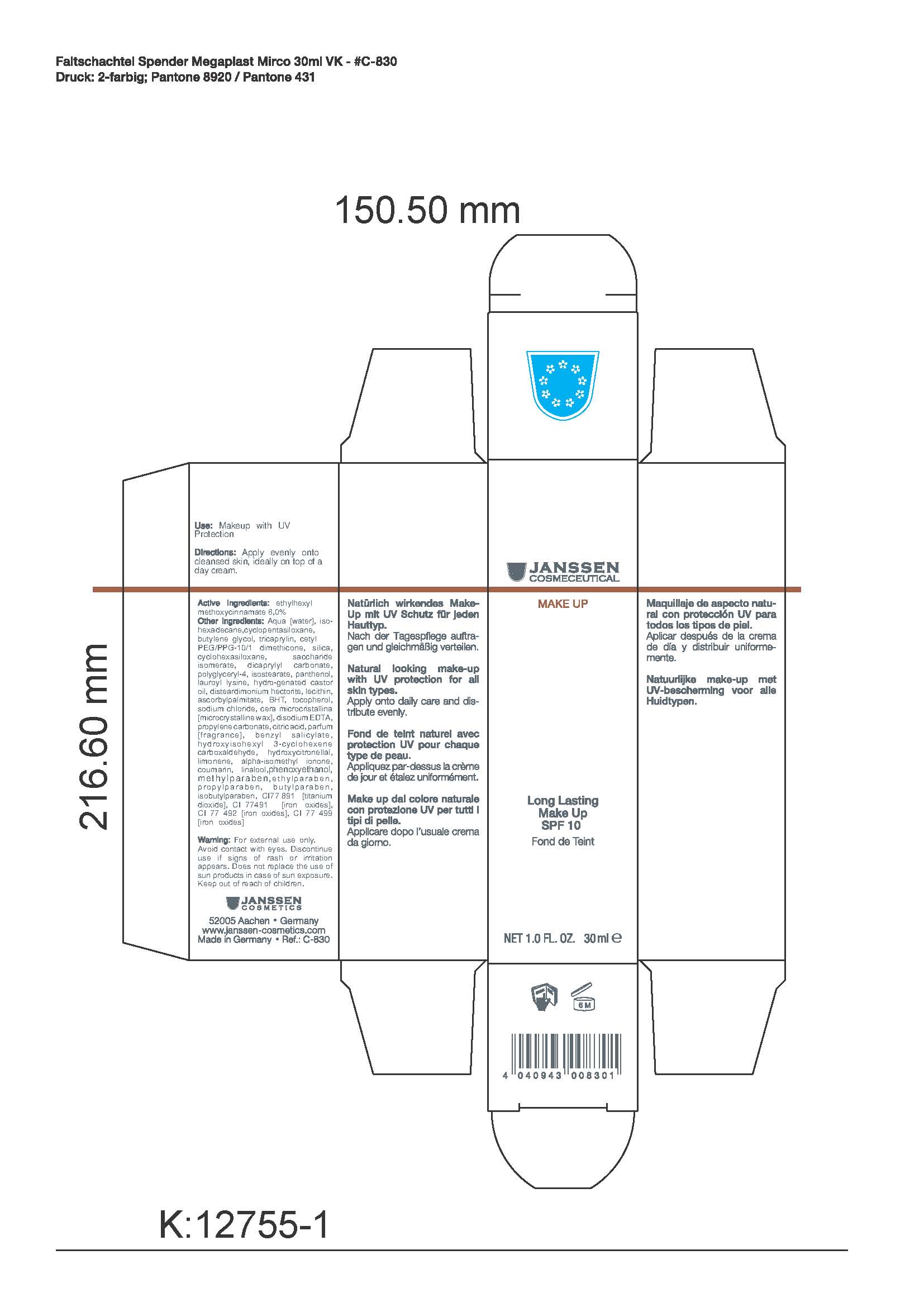
Inactive Ingredients: Aqua [water], isohexadecane, cyclopentasiloxane,butylene glycol, tricaprylin, cetyl PEG/PPG-10/1 dimethicone, silica, cyclohexasiloxane, saccharide isomerate, dicaprylyl carbonate, polyglyceryl-4, isostearate, panthenol, lauroyl lysine, hydro-genated castor oil, disteardimonium hectorite, lecithin, ascorbylpalmitate, BHT, tocopherol, sodium chloride, cera microcristallina [microcrystalline wax], disodium EDTA, propylene carbonate, citric acid, parfum [fragrance], benzyl salicylate, hydroxyisohexyl 3-cyclohexene carboxaldehyde, hydroxycitronellal, limonene, alpha-isomethyl ionone, coumarin, linalool,phenoxyethanol, methylparaben,ethylparaben, propylparaben, butylparaben,isobutylparaben, CI77 891 [titanium dioxide], CI 77491 [iron oxides],CI 77 492 [iron oxides], CI 77 499 [iron oxides]
- DO NOT USE
- KEEP OUT OF REACH OF CHILDREN
- PURPOSE
- STOP USE
- Directions
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LONG LAST MAKE UP 02 (NEO HELIOPAN)
ethylhexyl methoxycinnamate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:24653-805 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 6 mL in 30 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) ISOHEXADECANE (UNII: 918X1OUF1E) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) TRICAPRYLIN (UNII: 6P92858988) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) DIMETHICONE (UNII: 92RU3N3Y1O) Silicon Dioxide (UNII: ETJ7Z6XBU4) QUATERNIUM-18 (UNII: O7757NO1VL) HECTORITE (UNII: 08X4KI73EZ) SACCHARIDE ISOMERATE (UNII: W8K377W98I) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) POLYGLYCERYL-4 ISOSTEARATE (UNII: 820DPX33S7) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) LAUROYL LYSINE (UNII: 113171Q70B) PANTHENOL (UNII: WV9CM0O67Z) ASCORBYL PALMITATE (UNII: QN83US2B0N) ALPHA-TOCOPHEROL (UNII: H4N855PNZ1) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) EDETATE DISODIUM (UNII: 7FLD91C86K) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) SODIUM CHLORIDE (UNII: 451W47IQ8X) CITRIC ACID (UNII: 2968PHW8QP) PROPYLENE CARBONATE (UNII: 8D08K3S51E) BENZYL SALICYLATE (UNII: WAO5MNK9TU) HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (UNII: QUE43B9Z2Q) HYDROXYCITRONELLAL (UNII: 8SQ0VA4YUR) LIMONENE, (+)- (UNII: GFD7C86Q1W) IONONE (UNII: QP734LIN1K) COUMARIN (UNII: A4VZ22K1WT) LINALOOL, DL- (UNII: D81QY6I88E) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) ETHYLPARABEN (UNII: 14255EXE39) PROPYLPARABEN (UNII: Z8IX2SC1OH) BUTYLPARABEN (UNII: 3QPI1U3FV8) ISOBUTYLPARABEN (UNII: 0QQJ25X58G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24653-805-02 1 in 1 CARTON 1 NDC:24653-805-01 30 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 10/10/2008 Labeler - Janssen Cosmetics GmbH (499187946) Registrant - Janssen Cosmeceutical LLC (052993438) Establishment Name Address ID/FEI Business Operations Janssen Cosmecetics GmbH 499187946 manufacture Establishment Name Address ID/FEI Business Operations Dr. Sacher Kosmetik GmbH 341926681 api manufacture