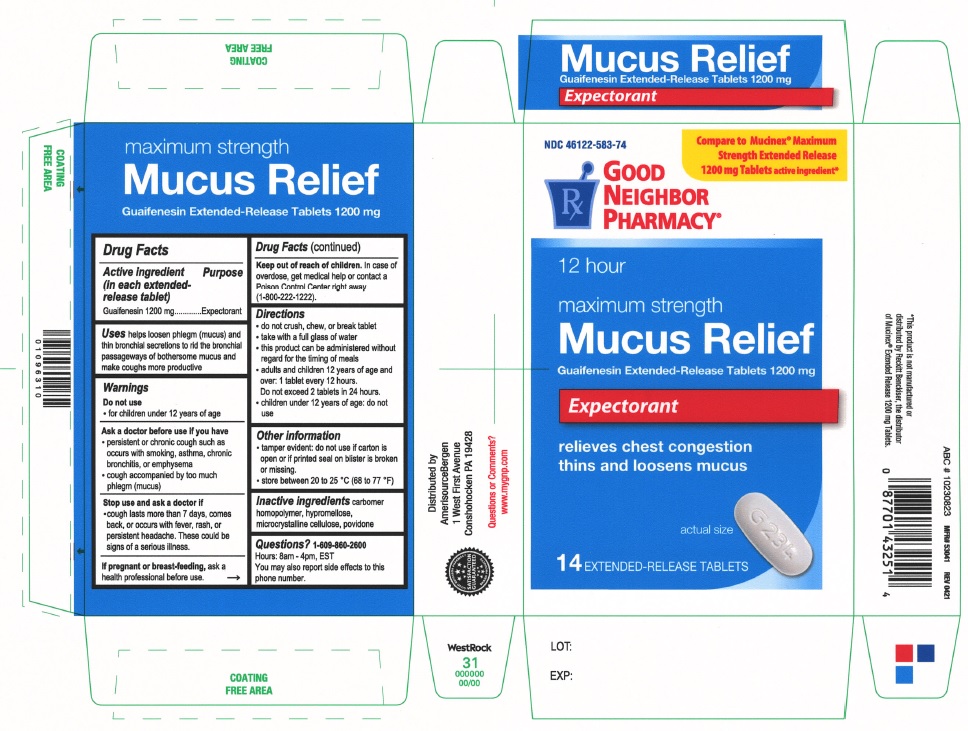
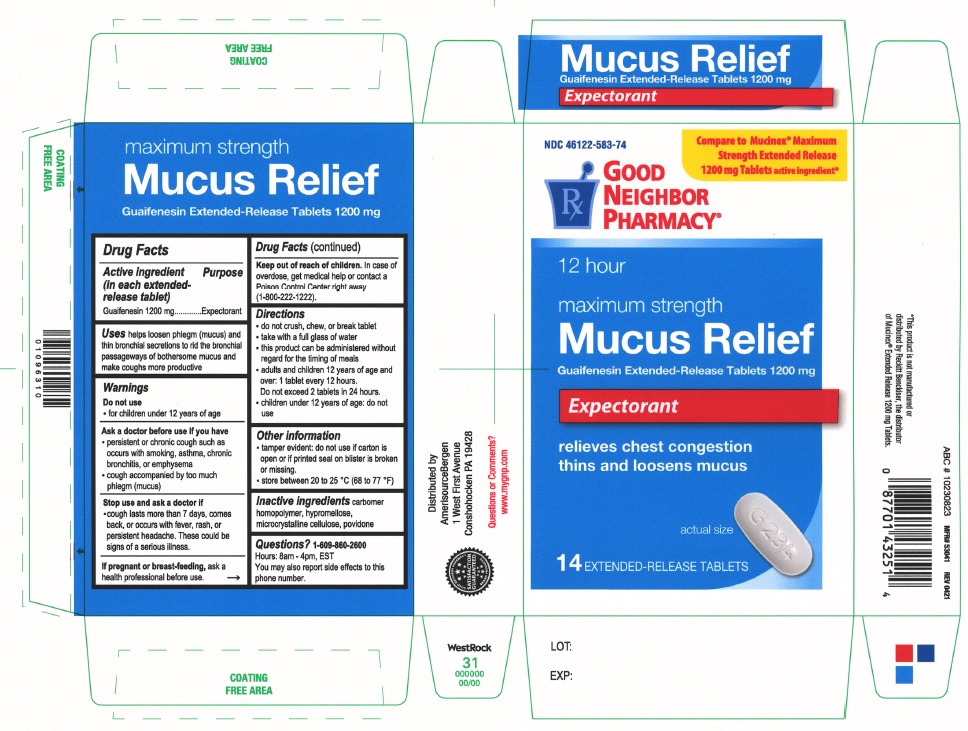
Label: MAXIMUM STRENGTH MUCUS RELIEF- guaifenesin tablet, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 46122-583-74 - Packager: Amerisource Bergen
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 11, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT(in each extended-release tablet)
- PURPOSE
- USE(S)
- WARNING
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- STOP USE AND ASK A DOCTOR IF
- IF PREGNANT OR BREAST-FEEDING,
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS?
-
PRINCIPAL DISPLAY PANEL
Compare to Mucinex® Maximum Strength Extended Release 1200 mg Tablets active ingredient
NDC 46122-583-74
Good Neighbor Pharmacy
12 hour
maximum strength
Mucus ReliefGuaifenesin Extended-Release Tablets 1200 mg
Expectorant
relieves chest congestion
thins and loosens mucus
14 EXTENDED-RELEASE TABLETS
-
INGREDIENTS AND APPEARANCE
MAXIMUM STRENGTH MUCUS RELIEF
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46122-583 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) HYPROMELLOSE 2208 (15000 MPA.S) (UNII: Z78RG6M2N2) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 22mm Flavor Imprint Code G234 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46122-583-74 2 in 1 CARTON 11/01/2019 1 7 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209215 11/01/2019 Labeler - Amerisource Bergen (007914906)