Label: GOLD MOUNTAIN BEAUTY FUNGAL NAIL ELIMINATOR- tolnaftate antifungal oil
-
Contains inactivated NDC Code(s)
NDC Code(s): 76348-417-01, 76348-417-02 - Packager: Renu Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 14, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- For external use only
- KEEP OUT OF REACH OF CHILDREN
- OTHER SAFETY INFORMATION
- Inactive ingredients
- ASK DOCTOR
- QUESTIONS
- STOP USE
-
INDICATIONS & USAGE
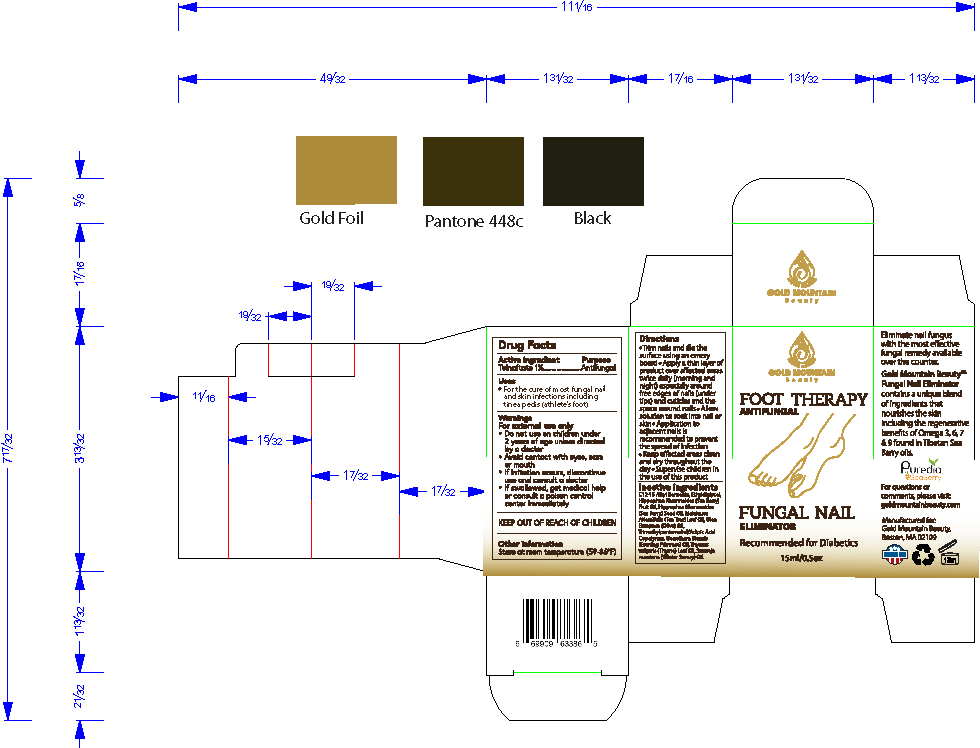
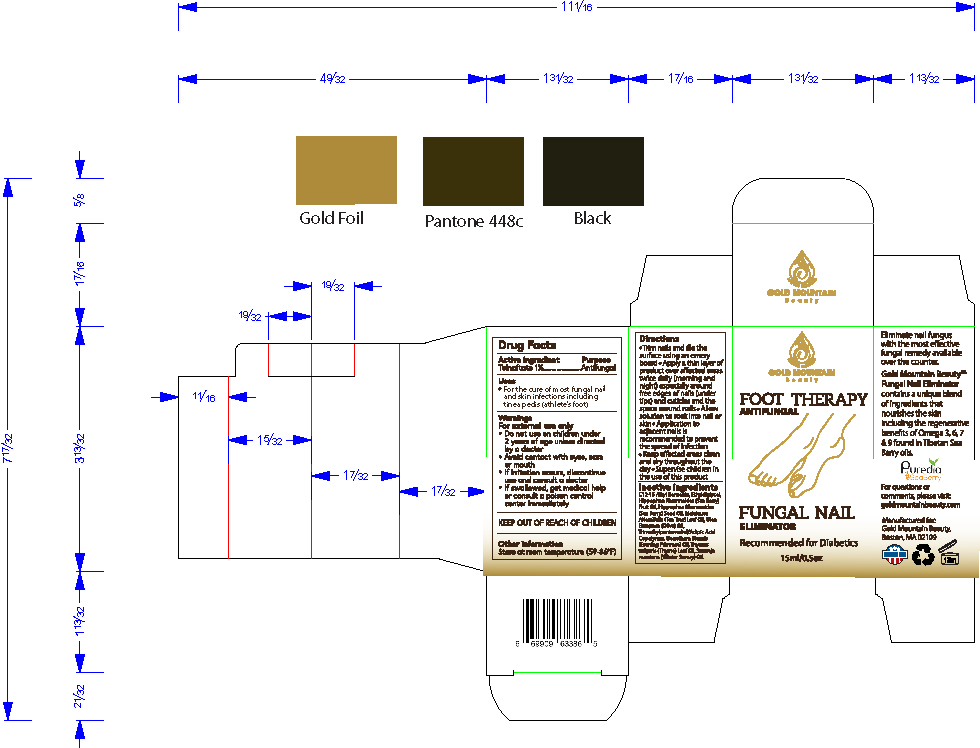
Trim nails and file the surface using an emery board
Apply a thin layer of product over affected areas twice daily (morning and night) especially around free edges of nails (under tips) and cuticles and the space around nails
Allow solution to soak into nail or skin
Application to adjacent nails is recommended to prevent the spread of infection
Keep affected areas clean and dry throughout the day
Supervise children in the use of this product - WARNINGS
-
DOSAGE & ADMINISTRATION
Directions
- Trim nails and file the surface using an emery board
- Apply a thin layer of product over affected areas twice daily (morning and night) especially around free edges of nails (under tips) and cuticles and the space around nails
- Allow solution to soak into nail or skin
- Application to adjacent nails is recommended to prevent the spread of infection
- Keep affected areas clean and dry throughout the day
- Supervise children in the use of this product
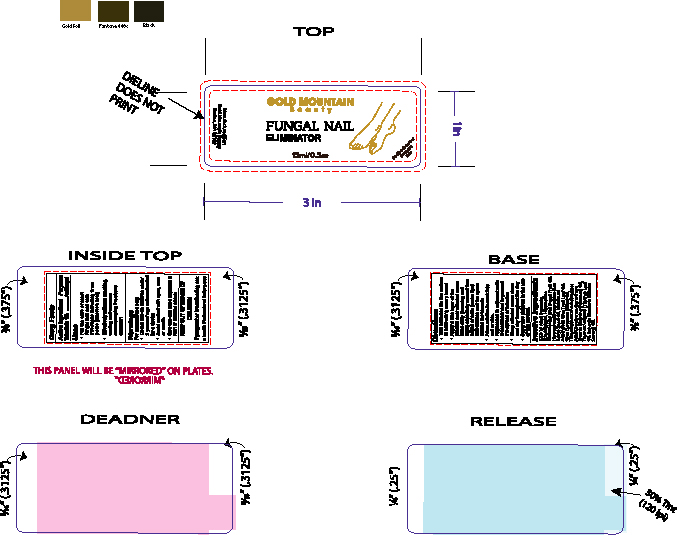
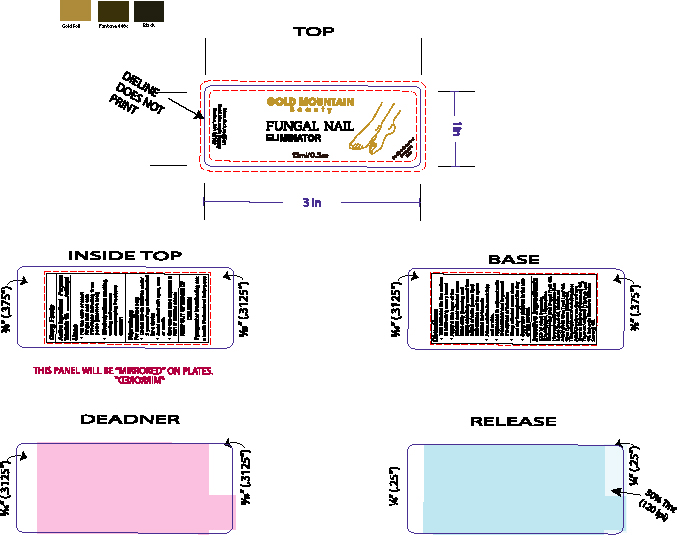
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GOLD MOUNTAIN BEAUTY FUNGAL NAIL ELIMINATOR
tolnaftate antifungal oilProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76348-417 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 0.14 g in 14 g Inactive Ingredients Ingredient Name Strength THYME OIL (UNII: 2UK410MY6B) EVENING PRIMROSE OIL (UNII: 3Q9L08K71N) WINTER SAVORY OIL (UNII: EP61SM69Z2) TRIMETHYLPENTANEDIOL/ADIPIC ACID/GLYCERIN CROSSPOLYMER (25000 MPA.S) (UNII: 587WKM3S9Q) OLIVE OIL (UNII: 6UYK2W1W1E) HIPPOPHAE RHAMNOIDES FRUIT OIL (UNII: TA4JCF9S1J) DIETHYLENE GLYCOL MONOETHYL ETHER (UNII: A1A1I8X02B) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) TEA TREE OIL (UNII: VIF565UC2G) HIPPOPHAE RHAMNOIDES SEED OIL (UNII: T53SBG6741) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76348-417-02 1 in 1 BOX 07/02/2018 1 NDC:76348-417-01 14 g in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 07/02/2018 Labeler - Renu Laboratories, Inc. (945739449) Establishment Name Address ID/FEI Business Operations Renu Laboratories Inc. 945739449 manufacture(76348-417)

 GOLD MOUNTAIN
GOLD MOUNTAIN