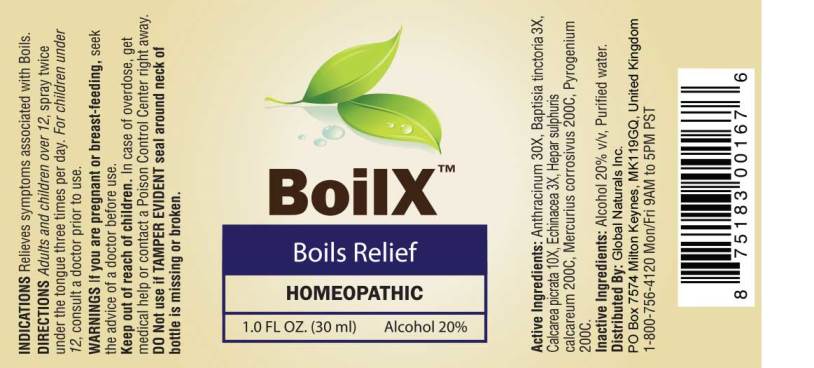
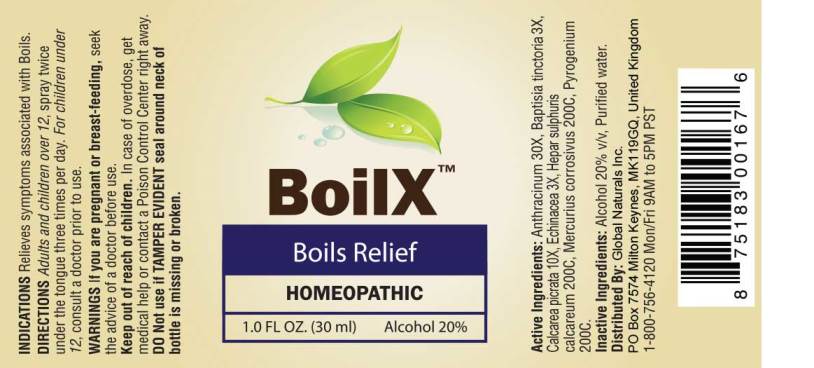
Label: BOILX (anthracinum, baptisia tinctoria, calcarea picrata, echinacea- angustifolia, hepar sulphuris calcareum, mercurius corrosivus, pyrogenium spray
- NDC Code(s): 43695-0012-1
- Packager: Global Naturals, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated June 1, 2018
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS:
- INDICATIONS:
- WARNINGS:
- KEEP OUT OF REACH OF CHILDREN:
- DIRECTIONS:
- INDICATIONS:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
BOILX
anthracinum, baptisia tinctoria, calcarea picrata, echinacea (angustifolia), hepar sulphuris calcareum, mercurius corrosivus, pyrogenium sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43695-0012 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACILLUS ANTHRACIS IMMUNOSERUM RABBIT (UNII: 41LZ22DD4N) (BACILLUS ANTHRACIS IMMUNOSERUM RABBIT - UNII:41LZ22DD4N) BACILLUS ANTHRACIS IMMUNOSERUM RABBIT 30 [hp_X] in 1 mL BAPTISIA TINCTORIA ROOT (UNII: 5EF0HWI5WU) (BAPTISIA TINCTORIA ROOT - UNII:5EF0HWI5WU) BAPTISIA TINCTORIA ROOT 3 [hp_X] in 1 mL CALCIUM PICRATE (UNII: 53D441QVT8) (CALCIUM PICRATE - UNII:53D441QVT8) CALCIUM PICRATE 10 [hp_X] in 1 mL ECHINACEA ANGUSTIFOLIA (UNII: VB06AV5US8) (ECHINACEA ANGUSTIFOLIA - UNII:VB06AV5US8) ECHINACEA ANGUSTIFOLIA 3 [hp_X] in 1 mL CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 200 [hp_C] in 1 mL MERCURIC CHLORIDE (UNII: 53GH7MZT1R) (MERCURIC CATION - UNII:ED30FJ8Y42) MERCURIC CHLORIDE 200 [hp_C] in 1 mL RANCID BEEF (UNII: 29SUH5R3HU) (RANCID BEEF - UNII:29SUH5R3HU) RANCID BEEF 200 [hp_C] in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43695-0012-1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 06/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/01/2018 Labeler - Global Naturals, Inc. (184784283) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(43695-0012) , api manufacture(43695-0012) , label(43695-0012) , pack(43695-0012)