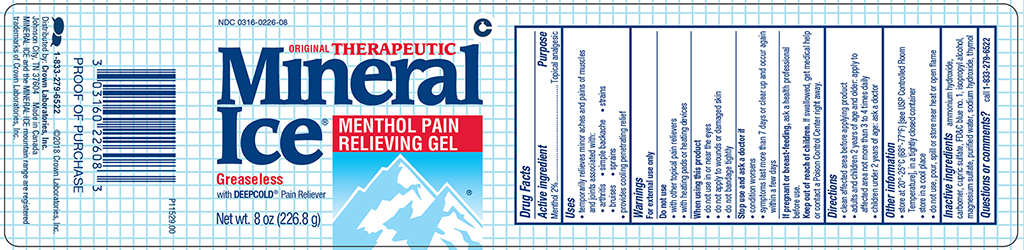
Label: MINERAL ICE PAIN RELIEVING- menthol gel
- NDC Code(s): 0316-0226-08, 0316-0226-16, 0316-0226-35
- Packager: Crown Laboratories
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Additional Information Listed on Other Panels
- Principal Display
-
INGREDIENTS AND APPEARANCE
MINERAL ICE PAIN RELIEVING
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0316-0226 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 20 mg in 1 g Inactive Ingredients Ingredient Name Strength AMMONIA (UNII: 5138Q19F1X) CARBOMER HOMOPOLYMER TYPE B (UNII: HHT01ZNK31) CUPRIC SULFATE (UNII: LRX7AJ16DT) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) ISOPROPYL ALCOHOL (UNII: ND2M416302) MAGNESIUM SULFATE HEPTAHYDRATE (UNII: SK47B8698T) SODIUM HYDROXIDE (UNII: 55X04QC32I) THYMOL (UNII: 3J50XA376E) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0316-0226-35 99.2 g in 1 JAR; Type 0: Not a Combination Product 12/01/2018 2 NDC:0316-0226-08 226.8 g in 1 JAR; Type 0: Not a Combination Product 12/01/2018 3 NDC:0316-0226-16 453.6 g in 1 JAR; Type 0: Not a Combination Product 12/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 11/01/2011 Labeler - Crown Laboratories (079035945)