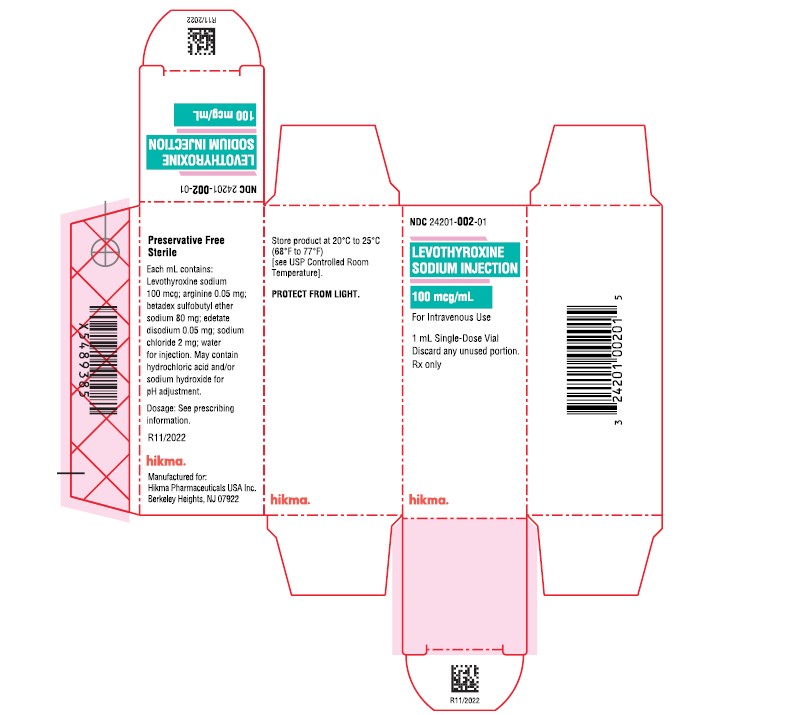
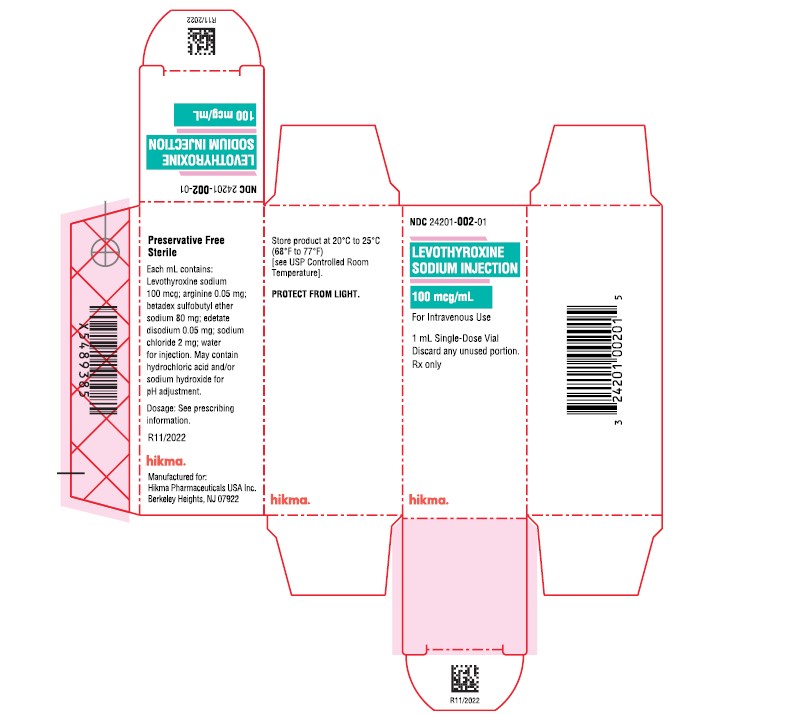
Label: LEVOTHYROXINE SODIUM injection, solution
- NDC Code(s): 24201-002-01
- Packager: Hikma Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 1, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LEVOTHYROXINE SODIUM INJECTION safely and effectively. See full prescribing information for LEVOTHYROXINE SODIUM INJECTION
LEVOTHYROXINE SODIUM injection, for intravenous use
Initial U.S. Approval: 1969WARNING: NOT FOR TREATMENT OF OBESITY OR FOR WEIGHT LOSS
Thyroid hormones, including Levothyroxine Sodium Injection, should not be used for the treatment of obesity or for weight loss. (5.3) Larger doses may produce serious or even life-threatening manifestations of toxicity. (6)
INDICATIONS AND USAGE
Levothyroxine Sodium is an L-thyroxine product. Levothyroxine (T4) Sodium Injection is indicated for the treatment of myxedema coma. (1) (1)
Important Limitations of Use: (1)
The relative bioavailability of this drug has not been established. Use caution when converting patients from oral to intravenous levothyroxine. (1) (1)
DOSAGE AND ADMINISTRATION
- Consider the patient’s age, general physical condition, cardiac risk factors, as well as the clinical severity of myxedema and duration of myxedema symptoms when determining dosages of Levothyroxine Sodium Injection. (2.1)
- Start with lower doses in elderly patients and in patients with underlying cardiovascular disease. (2.1)
- An initial intravenous loading dose of Levothyroxine Sodium Injection between 300 to 500 mcg followed by once daily intravenous maintenance doses between 50 and 100 mcg should be administered, as clinically indicated, until the patient can tolerate oral therapy. (2.1)
- Do not add to other IV fluids. (2.2)
DOSAGE FORMS AND STRENGTHS
Injection: 100 mcg/mL in a single-dose vial. Ready-to-use solution. (3) (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
- Cardiac Adverse Reactions in the Elderly and in Patients with Underlying Cardiovascular Disease: Overtreatment may cause arrhythmias, tachycardia, myocardial ischemia and infarction, or worsening of congestive heart failure and death, particularly in patients with cardiovascular disease and in elderly patients. Start with lower doses in elderly patients and in patients with underlying cardiovascular disease and monitor patients after administration (5.1).
- Acute Adrenal Crisis in Patients with Concomitant Adrenal Insufficiency Initiation of thyroid hormone therapy prior to initiating glucocorticoid therapy may precipitate an acute adrenal crisis in patients with adrenal insufficiency. Treat patients with adrenal insufficiency with replacement glucocorticoids prior to initiating treatment (5.2).
- Worsening of Diabetic Control: May worsen glycemic control and result in increased antidiabetic agent or insulin requirements. Carefully monitor glycemic control (5.3)
ADVERSE REACTIONS
Adverse reactions associated with Levothyroxine Sodium Injection are primarily those of hyperthyroidism due to therapeutic overdosage: fatigue, increased appetite, weight loss, heat intolerance, fever, excessive sweating, headache, hyperactivity, nervousness, anxiety, irritability, emotional lability, insomnia, tremors, muscle weakness, muscle spasm, palpitations, tachycardia, arrhythmias, increased pulse and blood pressure, heart failure, angina, myocardial infarction, cardiac arrest, dyspnea, diarrhea, vomiting, abdominal cramps, elevations in liver function tests, flushing, and rash. (6) (6)
To report SUSPECTED ADVERSE REACTIONS, contact Hikma at us.hikma@primevigilance.com or call 1-877-845-0689 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)
DRUG INTERACTIONS
Many drugs affect thyroid hormone pharmacokinetics and metabolism (e.g., absorption, synthesis, secretion, catabolism, protein binding, and target tissue response) and may alter the therapeutic response to Levothyroxine Sodium Injection. (7, 12.3) (7)
USE IN SPECIFIC POPULATIONS
Elderly and those with underlying cardiovascular disease should receive doses at the lower end of the recommended range. (8.5) (8)
Revised: 11/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: NOT FOR TREATMENT OF OBESITY OR FOR WEIGHT LOSS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
2.2 Dosing in the Elderly and in Patients with Cardiovascular Disease
2.3 Administration Instructions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Cardiac Complications in Elderly and in Patients with Cardiovascular Disease
5.2 Acute Adrenal Crisis in Patients with Concomitant Adrenal Insufficiency
5.3 Worsening of Diabetic Control
6 ADVERSE REACTIONS
7 DRUG INTERACTIONS
7.1 Drugs Known to Affect Thyroid Hormone Pharmacokinetics
7.2 Antidiabetic Therapy
7.3 Oral Anticoagulants
7.4 Digitalis Glycosides
7.5 Antidepressant Therapy
7.6 Ketamine
7.7 Sympathomimetics
7.8 Drug-Laboratory Test Interactions
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use and Patients with Underlying Cardiovascular Disease
8.6 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- BOXED WARNING (What is this?)
-
1 INDICATIONS AND USAGE
Levothyroxine Sodium Injection is indicated for the treatment of myxedema coma.
Limitations of Use: The relative bioavailability between Levothyroxine Sodium Injection and oral levothyroxine products has not been established. Caution should be used when switching patients from oral levothyroxine products to Levothyroxine Sodium Injection as accurate dosing conversion has not been studied.
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosage
An initial intravenous loading dose of Levothyroxine Sodium Injection between 300 to 500 mcg, followed by once daily intravenous maintenance doses between 50 and 100 mcg, should be administered, as clinically indicated, until the patient can tolerate oral therapy.
The age, general physical condition, and cardiac risk factors of the patient, as well as the clinical severity of myxedema and duration of myxedema symptoms should be considered when determining the starting and maintenance dosages of Levothyroxine Sodium Injection.
Levothyroxine Sodium Injection produces a gradual increase in the circulating concentrations of the hormone with an approximate half-life of 9 to 10 days in hypothyroid patients. Daily administration of Levothyroxine Sodium Injection should be maintained until the patient is capable of tolerating an oral dose and is clinically stable. For chronic treatment of hypothyroidism, an oral dosage form of levothyroxine should be used to maintain a euthyroid state. Relative bioavailability between Levothyroxine Sodium Injection and oral levothyroxine products has not been established. Based on medical practice, the relative bioavailability between oral and intravenous administration of Levothyroxine Sodium Injection is estimated to be from 48 to 74%. Due to differences in absorption characteristics of patients and the oral levothyroxine product formulations, TSH and thyroid hormone levels should be measured a few weeks after initiating oral levothyroxine and dose adjusted accordingly.
2.2 Dosing in the Elderly and in Patients with Cardiovascular Disease
Intravenous levothyroxine may be associated with cardiac toxicity – including arrhythmias, tachycardia, myocardial ischemia and infarction, or worsening of congestive heart failure and death – in the elderly and in those with underlying cardiovascular disease. Therefore, cautious use, including doses in the lower end of the recommended range, may be warranted in these populations.
2.3 Administration Instructions
Intravenous levothyroxine may be associated with cardiac toxicity-including arrhythmias, tachycardia, myocardial ischemia and infarction, or worsening of congestive heart failure and death—in the elderly and in those with underlying cardiovascular disease. Therefore, cautious use, including doses in the lower end of the recommended range, may be warranted in these populations.
Discard any unused portion. DO NOT ADD LEVOTHYROXINE SODIUM INJECTION TO OTHER INTRAVENOUS FLUIDS. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Cardiac Complications in Elderly and in Patients with Cardiovascular Disease
Excessive bolus dosing of Levothyroxine Sodium Injection (greater than 500 mcg) is associated with cardiac complications, particularly in the elderly and in patients with an underlying cardiac condition. Adverse events that can potentially be related to the administration of large doses of Levothyroxine Sodium Injection include arrhythmias, tachycardia, myocardial ischemia and infarction, or worsening of congestive heart failure and death. Cautious use, including doses in the lower end of the recommended range, may be warranted in these populations. Close observation of the patient following the administration of Levothyroxine Sodium Injection is advised.
5.2 Acute Adrenal Crisis in Patients with Concomitant Adrenal Insufficiency
Chronic autoimmune thyroiditis, which can lead to myxedema coma, may occur in association with other autoimmune disorders such as adrenal insufficiency, pernicious anemia, and insulin‑dependent diabetes mellitus. Patients should be treated with replacement glucocorticoids prior to initiation of treatment with Levothyroxine Sodium Injection, until adrenal function has been adequately assessed. Failure to do so may precipitate an acute adrenal crisis when thyroid hormone therapy is initiated, due to increased metabolic clearance of glucocorticoids by thyroid hormone. With initiation of Levothyroxine Sodium Injection, patients with myxedema coma should also be monitored for previously undiagnosed diabetes insipidus.
-
6 ADVERSE REACTIONS
Adverse reactions associated with levothyroxine are primarily those of hyperthyroidism due to therapeutic overdosage [see Warnings and Precautions (5), Overdosage (10)]. They include the following:
- General: fatigue, increased appetite, weight loss, heat intolerance, fever, excessive sweating
- Central nervous system: headache, hyperactivity, nervousness, anxiety, irritability, emotional lability, insomnia
- Musculoskeletal: tremors, muscle weakness, muscle spasm
- Cardiovascular: palpitations, tachycardia, arrhythmias, increased pulse and blood pressure, heart failure, angina, myocardial infarction, cardiac arrest
- Respiratory: dyspnea
- Gastrointestinal: diarrhea, vomiting, abdominal cramps, elevations in liver function tests
- Dermatologic: flushing, rash
Seizures have been reported rarely with the institution of levothyroxine therapy.
Hypersensitivity Reactions
Hypersensitivity reactions to inactive ingredients have occurred in patients treated with thyroid hormone products. These include urticaria, pruritus, skin rash, flushing, angioedema, various gastrointestinal symptoms (abdominal pain, nausea, vomiting and diarrhea), fever, arthralgia, serum sickness, and wheezing. Hypersensitivity to levothyroxine itself is not known to occur.
-
7 DRUG INTERACTIONS
7.1 Drugs Known to Affect Thyroid Hormone Pharmacokinetics
Many drugs affect thyroid hormone pharmacokinetics and metabolism (e.g., synthesis, secretion, catabolism, protein binding, and target tissue response) and may alter the therapeutic response to Levothyroxine Sodium Injection (see Tables 1-3).
Table 1: Drugs That May Alter T4 and Triiodothyronine (T3) Serum Transport Without Effecting Free Thyroxine (FT4) Concentration (Euthyroidism) Drug or Drug Class
Effect
Clofibrate
Estrogen-containing oral contraceptives
Estrogens (oral)
Heroin/Methadone
5-Fluorouracil
Mitotane
Tamoxifen
These drugs may increase serum thyroxine-binding globulin (TBG) concentration.
Androgens/Anabolic Steroids
Asparaginase
Glucocorticoids
Slow-Release Nictonic Acid
These drugs may decrease serum TBG concentration.
Potential impact (below): Administration of these agents with levothyroxine results in an initial transient increase in FT4. Continued administration results in a decrease in serum T4 and normal FT4 and TSH concentrations.
Salicylates (> 2 g/day)
Salicylates inhibit binding of T4 and T3 to TBG and transthyretin. An initial increase in serum FT4 is followed by return of FT4 to normal levels with sustained therapeutic serum salicylate concentrations, although total T4 levels may decrease by as much as 30%.
Other drugs:
Carbamazepine
Furosemide (> 80 mg IV)
Heparin
Hydantoins
Non-Steroidal Anti-Inflammatory Drugs
- Fenamates
These drugs may cause protein-binding site displacement. Furosemide has been shown to inhibit the protein binding of T4 to TBG and albumin, causing an increase free T4 fraction in serum. Furosemide competes for T4-binding sites on TBG, prealbumin, and albumin, so that a single high dose can acutely lower the total T4 level. Phenytoin and carbamazepine reduce serum protein binding of levothyroxine, and total and free T4 may be reduced by 20% to 40%, but most patients have normal serum TSH levels and are clinically euthyroid. Closely monitor thyroid hormone parameters.
Table 2: Drugs That May Alter Hepatic Metabolism of T4 (Hypothyroidism) Potential impact: Stimulation of hepatic microsomal drug-metabolizing enzyme activity may cause increased hepatic degradation of levothyroxine, resulting in increased levothyroxine requirements.
Drug or Drug Class
Effect
Phenobarbital
RifampinPhenobarbital has been shown to reduce the response to thyroxine. Phenobarbital increases L-thyroxine metabolism by inducing uridine 5’-diphospho-glucuronosyltransferase (UGT) and leads to a lower T4 serum levels. Changes in thyroid status may occur if barbiturates are added or withdrawn from patients being treated for hypothyroidism. Rifampin has been shown to accelerate the metabolism of levothyroxine.
Table 3: Drugs That May Decrease Conversion of T4 to T3 Potential impact: Administration of these enzyme inhibitors decreases the peripheral conversion of T4 to T3, leading to decreased T3 levels. However, serum T4 levels are usually normal but may occasionally be slightly increased.
Drug or Drug Class
Effect
Beta-adrenergic antagonists
e.g., Propranolol > 160 mg/day)
In patients treated with large doses of propranolol (> 160 mg/day), T3 and T4 levels change slightly, TSH levels remain normal, and patients are clinically euthyroid. It should be noted that actions of particular beta-adrenergic antagonists may be impaired when the hypothyroid patient is converted to the euthyroid state.
Glucocorticoids
(e.g., Dexamethasone ≥ 4 mg/day)Short-term administration of large doses of glucocorticoids may decrease serum T3 concentrations by 30% with minimal change in serum T4 levels. However, long-term glucocorticoid therapy may result in slightly decreased T3 and T4 levels due to decreased TBG production (See above).
Other drugs:
AmiodaroneAmiodarone inhibits peripheral conversion of levothyroxine (T4) to triiodothyronine (T3) and may cause isolated biochemical changes (increase in serum free-T4, and decreased or normal free-T3) in clinically euthyroid patients.
7.2 Antidiabetic Therapy
Addition of levothyroxine to antidiabetic or insulin therapy may result in increased antidiabetic agent or insulin requirements. Careful monitoring of diabetic control is recommended.
7.3 Oral Anticoagulants
Levothyroxine increases the response to oral anticoagulant therapy. Therefore, a decrease in the dose of anticoagulant may be warranted with correction of the hypothyroid. Closely monitor coagulation tests to permit appropriate and timely dosage adjustments.
7.4 Digitalis Glycosides
Levothyroxine may reduce the therapeutic effects of digitalis glycosides. Serum digitalis glycoside levels may be decreased when a hypothyroid patient becomes euthyroid, necessitating an increase in the dose of digitalis glycosides.
7.5 Antidepressant Therapy
Concurrent use of tricyclic (e.g., amitriptyline) or tetracyclic (e.g., maprotiline) antidepressants and levothyroxine may increase the therapeutic and toxic effects of both drugs, possibly due to increased receptor sensitivity to catecholamines. Toxic effects may include increased risk of cardiac arrhythmias and central nervous system stimulation. Levothyroxine may accelerate the onset of action of tricyclics. Administration of sertraline in patients stabilized on levothyroxine may result in increased levothyroxine requirements.
7.6 Ketamine
Concurrent use of ketamine and levothyroxine may produce marked hypertension and tachycardia. Closely monitor blood pressure and heart rate in these patients.
7.7 Sympathomimetics
Concurrent use may of sympathomimetics and levothyroxine may increase the effects of sympathomimetics or thyroid hormone. Thyroid hormones may increase the risk of coronary insufficiency when sympathomimetic agents are administered to patients with coronary artery disease.
7.8 Drug-Laboratory Test Interactions
Consider changes in TBG concentration when interpreting T4 and T3 values. Measure and evaluate unbound (free) hormone and/or determine the free T4 index (FT4I) in this circumstance. Pregnancy, infectious hepatitis, estrogens, estrogen containing oral contraceptives, and acute intermittent porphyria increase TBG concentrations. Nephrosis, severe hypoproteinemia, severe liver disease, acromegaly, androgens, and corticosteroids decrease TBG concentration. Familial hyper- or hypo-thyroxine binding globulinemias have been described, with the incidence of TBG deficiency approximating 1 in 9000.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The clinical data in pregnant women treated with oral levothyroxine to treat hypothyroidism do not indicate a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are no data available on the use of Levothyroxine Sodium Injection in pregnant women. There are risks to the mother and fetus associated with myxedema coma in pregnancy (see Clinical Considerations).
No developmental or reproductive toxicity studies in animals have been conducted with Levothyroxine Sodium Injection.
The estimated background risks of major birth defects and miscarriage for the indicated populations are unknown. All pregnancies have background risks of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Myxedema coma is a medical emergency that can be fatal if left untreated. Delaying treatment in pregnant women with myxedema coma increases the risk of maternal and fetal morbidity and mortality. Life-sustaining therapy for pregnant women with myxedema coma should not be withheld due to potential concerns regarding the effects of Levothyroxine Sodium Injection on the fetus.
8.2 Lactation
Risk Summary
Published studies report that levothyroxine is present in human milk following the administration of oral levothyroxine. No adverse effects on the breastfed infant have been reported, and there is no information on the effects of levothyroxine on milk production from levothyroxine oral treatment. There are no available data with use of Levothyroxine Sodium Injection in lactating women. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Levothyroxine Sodium Injection and any potential adverse effects on the breastfed infant from Levothyroxine Sodium Injection or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of Levothyroxine Sodium Injection have not been established in pediatric patients.
8.5 Geriatric Use and Patients with Underlying Cardiovascular Disease
See Section 2, Dosage and Administration, for full prescribing information in the geriatric patient population. Because of the increased prevalence of cardiovascular disease in the elderly, cautious use of Levothyroxine Sodium Injection in the elderly and in patients with known cardiac risk factors is advised. Atrial fibrillation is a common side effect associated with levothyroxine treatment in the elderly [see Dosage and Administration (2) and Warnings and Precautions (5)].
-
10 OVERDOSAGE
In general, the signs and symptoms of overdosage with levothyroxine are those of hyperthyroidism [see Warnings and Precautions (5) and Adverse Reactions (6)]. In addition, confusion and disorientation may occur. Cerebral embolism, shock, coma, and death have been reported. Excessive doses of Levothyroxine Sodium Injection (greater than 500 mcg) are associated with cardiac complications in patients with underlying cardiac disease.
Treatment of Overdosage
Levothyroxine Sodium Injection should be reduced in dose or temporarily discontinued if signs or symptoms of overdosage occur. To obtain up-to-date information about the treatment of overdose, a good resource is the certified Regional Poison Control Center. In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs, and unusual drug kinetics in the patient.
In the event of an overdose, appropriate supportive treatment should be initiated as dictated by the patient’s medical status.
-
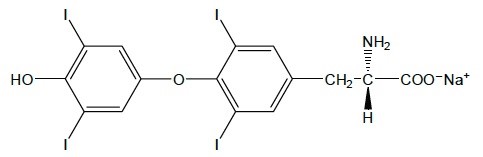
11 DESCRIPTION
Levothyroxine sodium injection contains synthetic levothyroxine (L-thyroxine) sodium salt. Levothyroxine sodium has an empirical formula of C15H10I4NNaO4, a molecular weight of 798.85 g/mol (anhydrous), and the following structural formula:
Levothyroxine sodium injection is a sterile, preservative-free, clear, colorless to slightly yellow solution for intravenous administration available as 100 mcg/mL in a single-dose clear glass vial. Each mL of levothyroxine sodium injection also contains 0.05 mg arginine, USP; 80 mg betadex sulfobutyl ether sodium, USP; 0.05 mg edetate disodium, USP; and water for injection, USP. Sodium chloride, USP was added to adjust tonicity. Hydrochloric acid, NF and/or sodium hydroxide, NF may have been added for pH adjustment.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Thyroid hormones exert their physiologic actions through control of DNA transcription and protein synthesis. Triiodothyronine (T3) and levothyroxine (T4) diffuse into the cell nucleus and bind to thyroid receptor proteins attached to DNA. This hormone nuclear receptor complex activates gene transcription and synthesis of messenger RNA and cytoplasmic proteins.
The physiological actions of thyroid hormones are produced predominantly by T3, the majority of which (approximately 80%) is derived from T4 by deiodination in peripheral tissues.
12.2 Pharmacodynamics
Thyroid hormone synthesis and secretion is regulated by the hypothalamic pituitary-thyroid axis. Thyrotropin releasing hormone (TRH) released from the hypothalamus stimulates secretion of thyrotropin stimulating hormone (TSH) from the anterior pituitary. TSH, in turn, is the physiologic stimulus for the synthesis and secretion of thyroid hormones, T4 and T3, by the thyroid gland. Circulating serum T3 and T4 levels exert a feedback effect on both TRH and TSH secretion. When serum T3 and T4 levels increase, TRH and TSH secretion decrease. When thyroid hormone levels decrease, TRH and TSH secretion increases. TSH is used for the diagnosis of hypothyroidism and evaluation of levothyroxine therapy adequacy with other laboratory and clinical data [see Dosage (2.1)]. There are drugs known to affect thyroid hormones and TSH by various mechanisms and those examples are diazepam, ethioamide, lovastatin, metoclopramide, 6-mercaptopurine, nitroprusside, perphenazine, and thiazide diuretics. Some drugs may cause a transient decrease in TSH secretion without hypothyroidism and those drugs (dose) are dopamine (greater than 1 mcg per kg per min), glucocorticoids (hydrocortisone greater than 100 mg per day or equivalent) and octreotide (greater than 100 mcg per day).
Thyroid hormones regulate multiple metabolic processes and play an essential role in normal growth and development, and normal maturation of the central nervous system and bone. The metabolic actions of thyroid hormones include augmentation of cellular respiration and thermogenesis, as well as metabolism of proteins, carbohydrates and lipids. The protein anabolic effects of thyroid hormones are essential to normal growth and development.
12.3 Pharmacokinetics
Absorption – Levothyroxine Sodium Injection is administered via the intravenous route. Following administration, the synthetic levothyroxine cannot be distinguished from the natural hormone that is secreted endogenously.
Distribution – Circulating thyroid hormones are greater than 99% bound to plasma proteins, including thyroxine binding globulin (TBG), thyroxine binding prealbumin (TBPA), and albumin (TBA), whose capacities and affinities vary for each hormone. The higher affinity of both TBG and TBPA for T4 partially explains the higher serum levels, slower metabolic clearance, and longer half-life of T4 compared to T3. Protein bound thyroid hormones exist in reverse equilibrium with small amounts of free hormone. Only unbound hormone is metabolically active. Many drugs and physiologic conditions affect the binding of thyroid hormones to serum proteins [see Warnings and Precautions (5) and Drug Interactions (7)]. Thyroid hormones do not readily cross the placental barrier [see Warnings and Precautions (5) and Use in Specific Populations (8)]. Betadex sulfobutyl ether (SBECD) does not affect levothyroxine binding to plasma proteins.
Metabolism – T4 is slowly eliminated. The major pathway of thyroid hormone metabolism is through sequential deiodination. Approximately eighty percent of circulating T3 is derived from peripheral T4 by mono-deiodination. The liver is the major site of degradation for both T4 and T3, with T4 deiodination also occurring at a number of additional sites, including the kidney and other tissues. Approximately 80% of the daily dose of T4 is deiodinated to yield equal amounts of T3 and reverse T3 (rT3). T3 and rT3 are further deiodinated to diiodothyronine. Thyroid hormones are also metabolized via conjugation with glucuronides and sulfates and excreted directly into the bile and gut where they undergo enterohepatic recirculation.
Elimination – Thyroid hormones are primarily eliminated by the kidneys. A portion of the conjugated hormone reaches the colon unchanged, where it is hydrolyzed and eliminated in feces as the free hormones. Urinary excretion of T4 decreases with age.
In patients with severe renal impairment or ESRD (eGFR of < 15 mL/min/1.73 m2), accumulation of SBECD occurs [see Renal Impairment 8.6].
Table 1: Pharmacokinetic Parameters of Thyroid Hormones in Euthyroid Patients
Hormone
Ratio in Thyroglobulin
Biologic Potency
Half-Life (Days)
Protein Binding
(%)2T4
10 to 20
1
6 to 81
99.96
T3
1
4
≤ 2
99.5
T4: Levothyroxine
T3: Liothyronine
13 to 4 days in hyperthyroidism, 9 to 10 days in hypothyroidism.
2Includes TBG, TBPA, and TBA. - 13 NONCLINICAL TOXICOLOGY
- 14 CLINICAL STUDIES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
Levothyroxine Sodium Injection 100 mcg/mL is a clear, colorless to slightly yellow solution, supplied as1 mL per vial.
Package of 1 single-dose vial: NDC 24201-002-01
16.2 Storage and Handling
Protect from light and store product at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Drug product is preservative free. Discard any unused portion.
Manufactured for:
Hikma Pharmaceuticals USA Inc.
Berkeley Heights, NJ 07922
www.hikma.com/us
Revised: 11/2022
INS-0017-R2
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LEVOTHYROXINE SODIUM
levothyroxine sodium injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24201-002 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEVOTHYROXINE SODIUM (UNII: 9J765S329G) (LEVOTHYROXINE - UNII:Q51BO43MG4) LEVOTHYROXINE SODIUM 100 ug in 1 mL Inactive Ingredients Ingredient Name Strength ARGININE (UNII: 94ZLA3W45F) EDETATE DISODIUM (UNII: 7FLD91C86K) BETADEX SULFOBUTYL ETHER SODIUM (UNII: 2PP9364507) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24201-002-01 1 in 1 CARTON 11/15/2022 1 2 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA214253 11/15/2022 Labeler - Hikma Pharmaceuticals USA Inc. (118707839) Registrant - Hikma Pharmaceuticals USA Inc. (946499746)