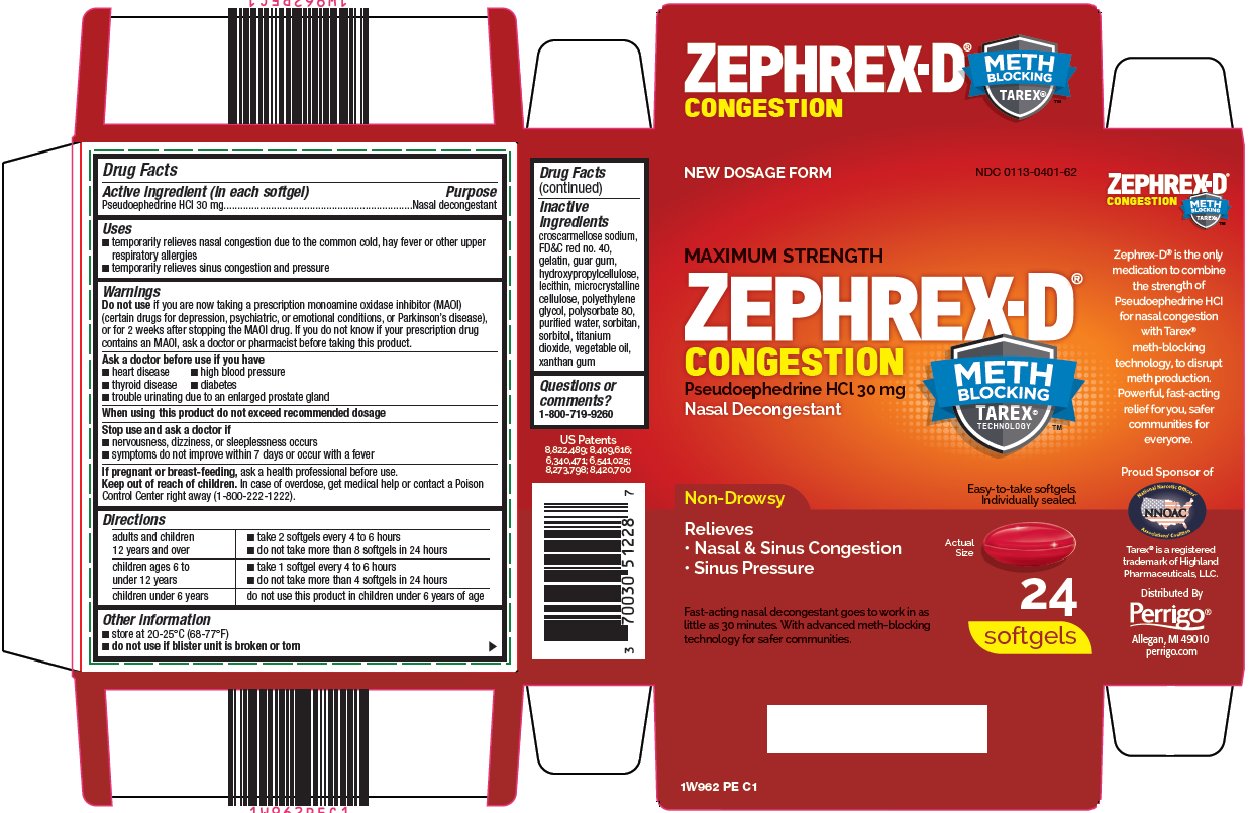
Label: ZEPHREX D- pseudoephedrine hydrochloride capsule, gelatin coated
- NDC Code(s): 0113-0401-62, 0113-0401-67
- Packager: L. Perrigo Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 18, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each softgel)
- Purpose
- Uses
- Warnings
-
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- Ask a doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions
adults and children 12 years and over
- •
- take 2 softgels every 4 to 6 hours
- •
- do not take more than 8 softgels in 24 hours
children ages 6 to under 12 years
- •
- take 1 softgel every 4 to 6 hours
- •
- do not take more than 4 softgels in 24 hours
children under 6 years
do not use this product in children under 6 years of age
- Other information
- Inactive ingredients
- Questions or comments?
-
Package/Label Principal Display Panel
NEW DOSAGE FORM
MAXIMUM STRENGTH
ZEPHREX-D ®
CONGESTION
Pseudoephedrine HCl 30 mg
Nasal Decongestant
METH BLOCKING TAREX® TECHNOLOGY ™
Non-Drowsy
Relieves
Nasal & Sinus Congestion
Sinus Pressure
Easy-to-take softgels.
Individually sealed.
Actual Size
Fast-acting nasal decongestant goes to work in as little as 30 minutes. With advanced meth-blocking technology for safer communities.
24 softgels
-
INGREDIENTS AND APPEARANCE
ZEPHREX D
pseudoephedrine hydrochloride capsule, gelatin coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0113-0401 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PSEUDOEPHEDRINE HYDROCHLORIDE (UNII: 6V9V2RYJ8N) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE HYDROCHLORIDE 30 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GUAR GUM (UNII: E89I1637KE) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) WATER (UNII: 059QF0KO0R) SORBITAN (UNII: 6O92ICV9RU) SORBITOL (UNII: 506T60A25R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color RED Score no score Shape OVAL Size 17mm Flavor Imprint Code ZD3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0113-0401-62 24 in 1 CARTON 04/17/2017 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:0113-0401-67 48 in 1 CARTON 05/08/2017 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 11/04/2016 Labeler - L. Perrigo Company (006013346)