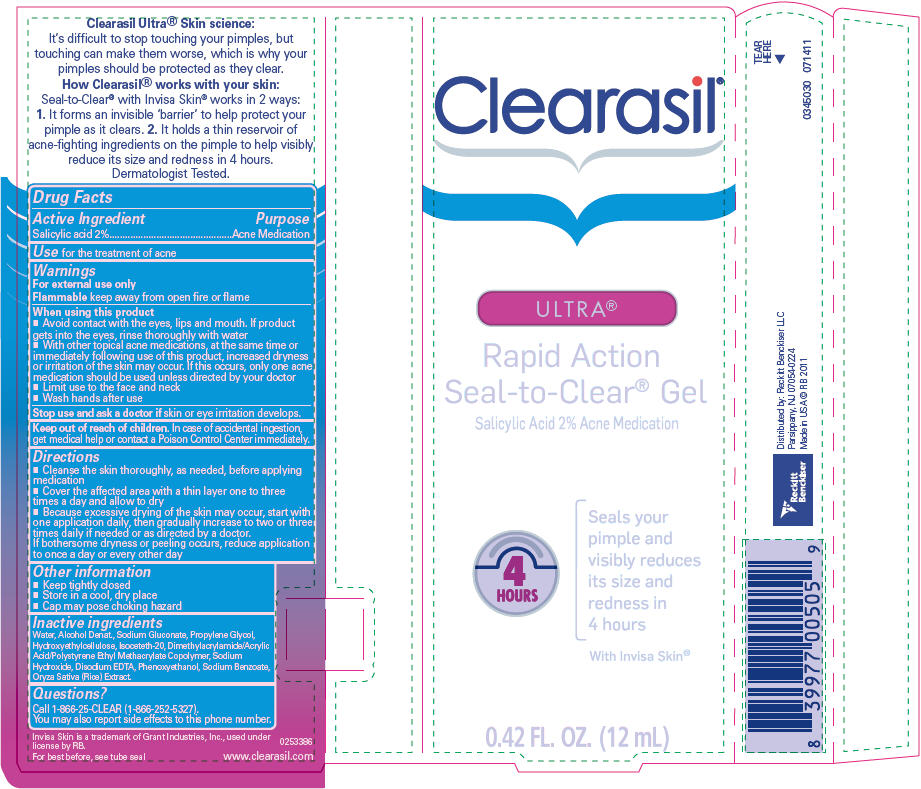
Label: CLEARASIL ULTRA RAPID ACTION SEAL-TO-CLEAR- salicylic acid gel
- NDC Code(s): 63824-353-01
- Packager: RB Health (US) LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Use
-
Warnings
For external use only
When using this product
- Avoid contact with the eyes, lips and mouth. If product gets into the eyes, rinse thoroughly with water
- With other topical acne medications, at the same time or immediately following use of this product, increased dryness or irritation of the skin may occur. If this occurs, only one acne medication should be used unless directed by your doctor
- Limit use to the face and neck
- Wash hands after use
-
Directions
- Cleanse the skin thoroughly, as needed, before applying medication
- Cover the affected area with a thin layer one to three times a day and allow to dry
- Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor.
If bothersome dryness or peeling occurs, reduce application to once a day or every other day
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 12 mL Tube Label
-
INGREDIENTS AND APPEARANCE
CLEARASIL ULTRA RAPID ACTION SEAL-TO-CLEAR
salicylic acid gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63824-353 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 2 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM GLUCONATE (UNII: R6Q3791S76) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ISOCETETH-20 (UNII: O020065R7Z) SODIUM HYDROXIDE (UNII: 55X04QC32I) EDETATE DISODIUM (UNII: 7FLD91C86K) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM BENZOATE (UNII: OJ245FE5EU) RICE OIL (UNII: ACU8G2V809) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63824-353-01 1 in 1 CONTAINER 07/21/2011 09/01/2024 1 12 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 07/21/2011 09/01/2024 Labeler - RB Health (US) LLC (081049410)