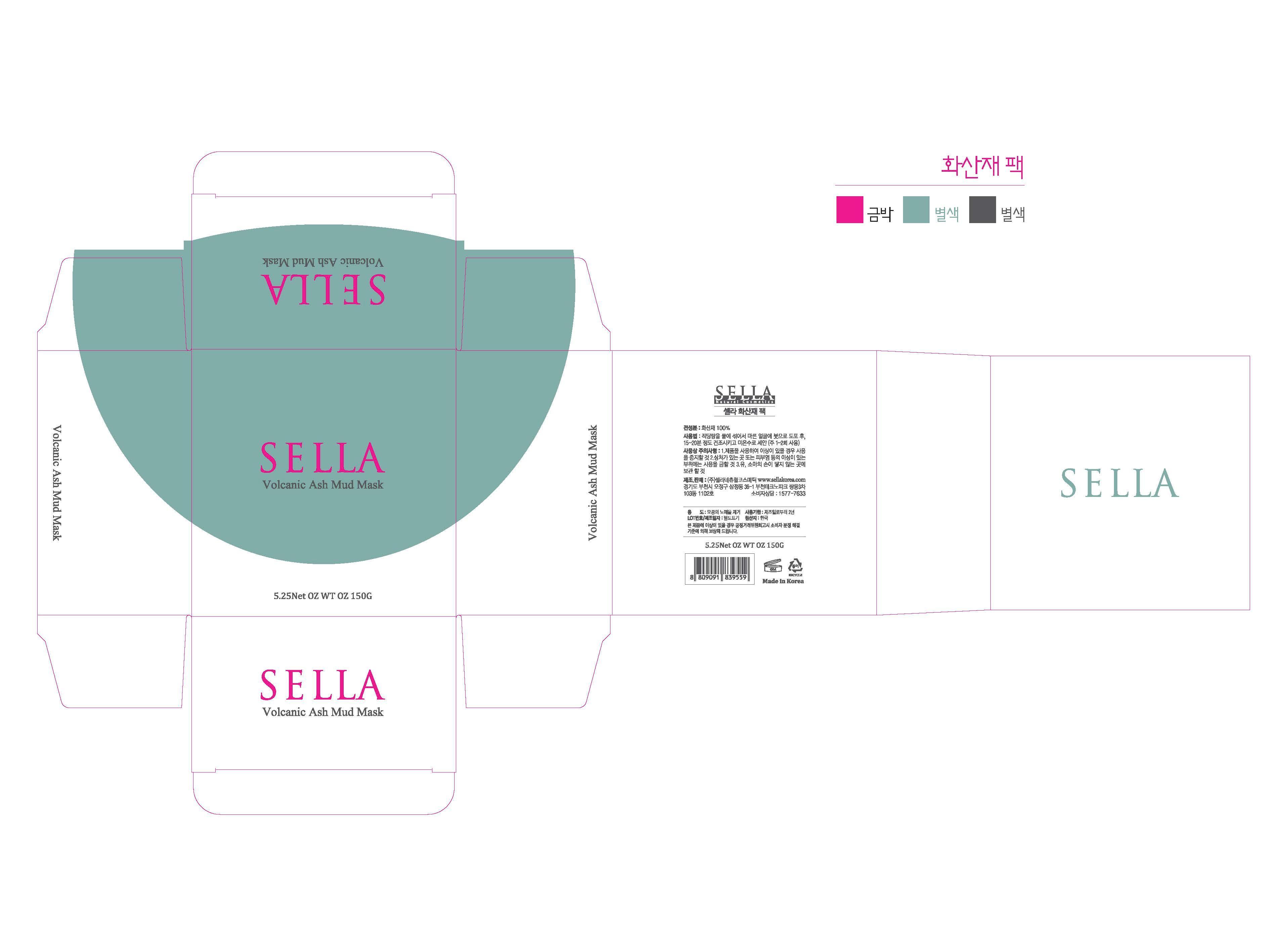
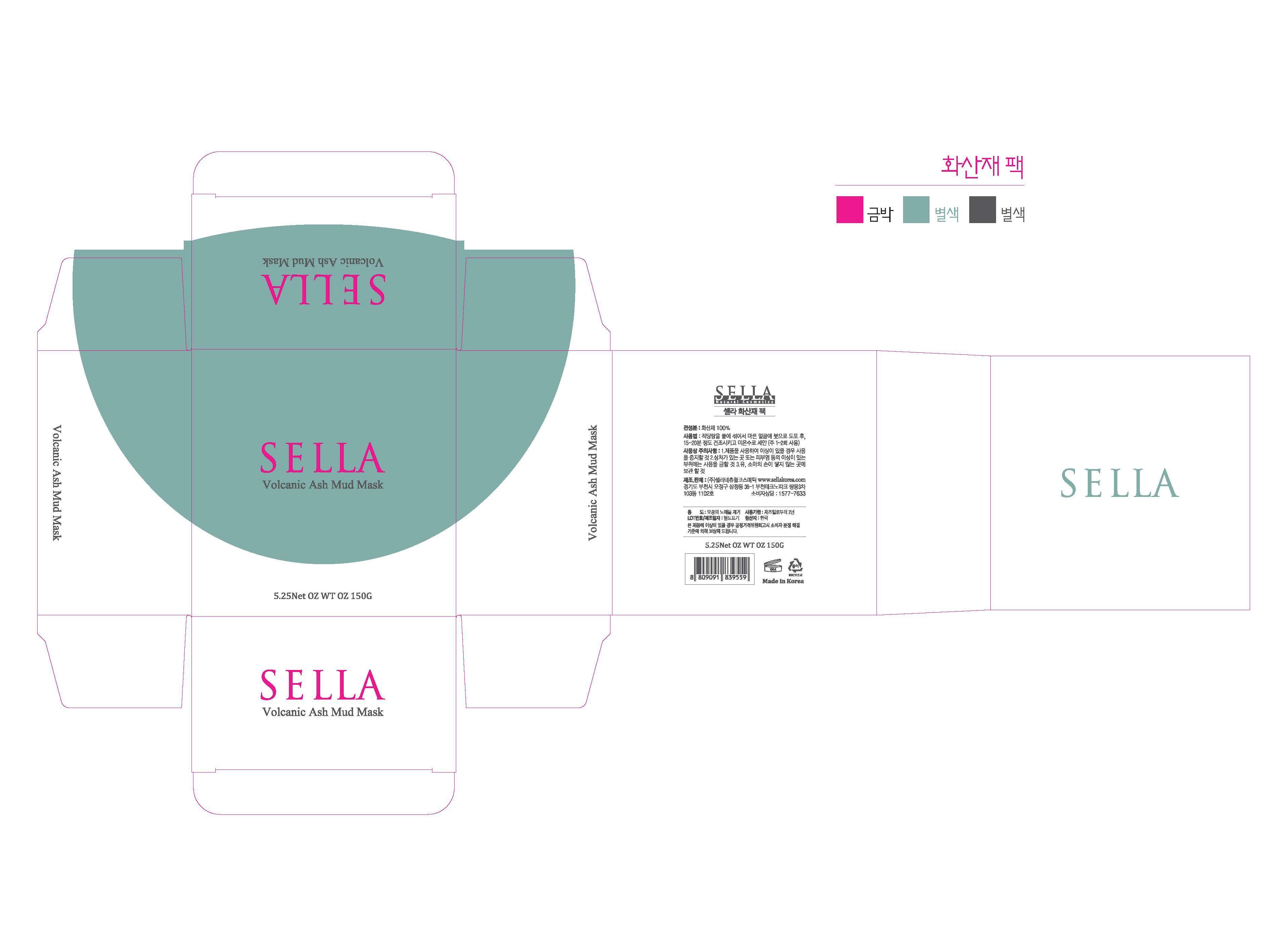
Label: SELLA NATURAL VOLCANIC ASH MUD MASK- silanol powder
-
Contains inactivated NDC Code(s)
NDC Code(s): 69603-1301-1 - Packager: Sella Natural Cosmetics Co.,Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 11, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
1. place on spoon ful of the volcanic ash powder in the mixing container
2. add purified water until it the water level reaches the line
3. get the lid on the mixing container and shake to mix well
4. wash face with sella cheansing bar three times
5. dry for 20 to 25 minutes, then wash off with warm water
6. apply sell gold serum and essence cream
-
WARNINGS
・Stop using the product when you have skin problems or the product disagrees with your skin
・Stop using the product immediately and consult a dermatologist if you have redness, swelling, itching or irritation on the skin while or after using the product.
・Don't place the product in any place where it will be subjected to extremely high or low temperatures or direct sunlight.
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SELLA NATURAL VOLCANIC ASH MUD MASK
silanol powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69603-1301 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILANOL (UNII: 079V3J9O3X) (SILANOL - UNII:079V3J9O3X) SILANOL 34.1 g in 100 g Inactive Ingredients Ingredient Name Strength ALUMINUM OXIDE (UNII: LMI26O6933) SODIUM OXIDE (UNII: 3075U8R23D) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69603-1301-1 150 g in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 02/11/2015 Labeler - Sella Natural Cosmetics Co.,Ltd. (687563333) Registrant - Sella Natural Cosmetics Co.,Ltd. (687563333) Establishment Name Address ID/FEI Business Operations Sella Natural Cosmetics Co.,Ltd. 687563333 manufacture(69603-1301)