Label: GLEPTOFORTE- gleptoferron injection
- NDC Code(s): 13744-525-01, 13744-525-02
- Packager: Ceva Sante Animale
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated December 20, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
GleptoForte®
(gleptoferron)
200 mg/mL of iron
Solution for intramuscular injection
NOT FOR USE IN HUMANS
KEEP OUT OF REACH OF CHILDREN
Approved by FDA under ANADA # 200-587
DESCRIPTION: GleptoForte® is a sterile aqueous colloidal solution of gleptoferron, a macromolecular complex of ferric hydroxide and dextran glucoheptonic acid. Each milliliter contains 200 mg of elemental iron. The solution contains 0.5% phenol as preservative.
-
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION: GleptoForte® is for intramuscular use only. Prevention: 1 mL (200 mg iron) per pig on or before 3 days of age. Treatment: 1 mL (200 mg iron) per pig as soon as signs of deficiency appear.
WITHDRAWAL PERIODS: No withdrawal period is required when used according to labeling.
ANIMAL SAFETY WARNINGS AND PRECAUTIONS: Occasionally pigs may show a reaction to injectable iron, clinically characterized by prostration with muscular weakness. In extreme cases, death may occur.
For a copy of the Safety Data Sheet (SDS) or to report suspected adverse drug events, contact CEVA at 1-800-999-0297.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/AnimalVeterinary/SafetyHealth.
HOW TO INJECT:
What to use
1. GleptoForte® (gleptoferron) injection
2. Ordinary 10 mL syringe with 18-guage needle or smaller, or dosage delivery device with 4-guage draw off spike or smaller
3. Disinfectant (rubbing alcohol or tincture of iodine)
Where to use:
GleptoForte® is for intramuscular use. The preferred site of injection is the ham muscle. Push the needle directly into the ham to approximately a one-half (1/2) inch depth. Avoid injection into a blood vessel. This is best avoided by pulling back slightly on the plunger after the needle is seated in the ham. If properly located, resistance will be offered to this effort. If the plunger is pulled back easily, remove and relocate the needle.
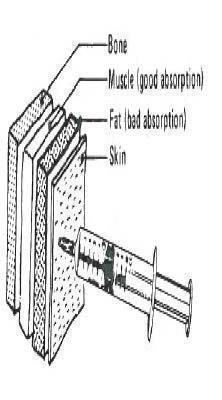
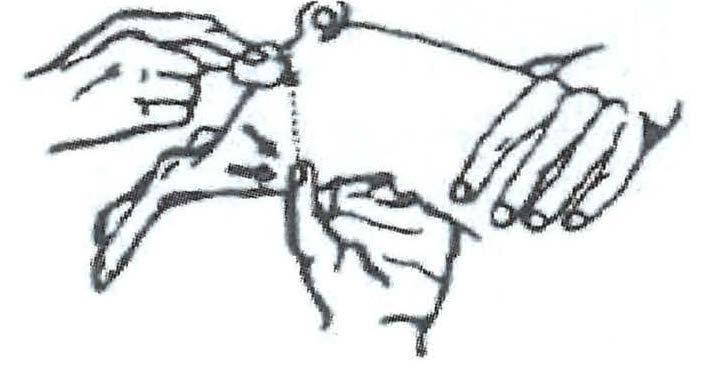
Step 1 The pig is held under-arm, between knees, or by an assistant. Disinfect the injection site. Pull down skin and fat with thumb as shown. Approximately halfway along a line between the anus and the thumb is the best injection site.
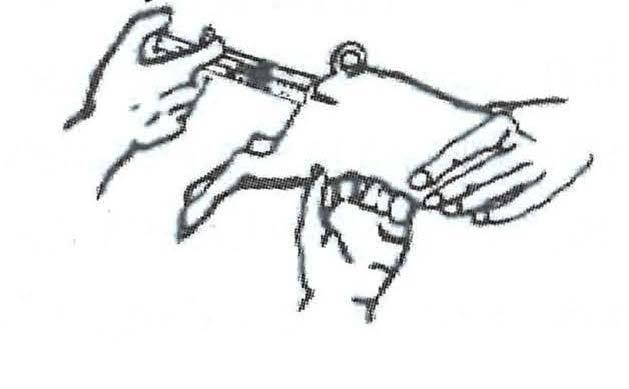
Step 2 Before injecting GleptoForte®, point the needle upward and gently press the plunger to expel all air from syringe. The needle is inserted at right-angle to the skin into the ham approximately a one-half (1/2) inch depth and the injection made.
Step 3 Use a new needle between each stopper puncture. Protect the needle from contamination and disinfect between animal injections.
- HOW SUPPLIED
-
STORAGE AND HANDLING
STORAGE: Store at 20°C - 25°C (68°F - 77°F). Do not freeze.
100 mL Vial: Use within 28 days of first puncture and puncture a maximum of seventy-five (75) times with an 18-guage needle or five (5) times with a dosage delivery device. If using a needle larger than 18-guage or a draw-off spike larger than 4-guage, discard any remaining product immediately ater use.
250 mL Vial: Use within 28 days of first puncture and puncture a maximum of one hundred (100) times with an 18-guage needle or five (5) times with a dosage delivery device. If using a needle larger than 18-guage or a draw-off spike larger than 4-guage, discard any remaining product immediately ater use.
Manufactured for:
Ceva Animal Health, LLC, Lenexa, KS 66215
1-800-999-0297GleptoForte® is a registered trademark of Ceva Santé Animale
- Principal Display Panel - 100 mL Bottle Label
- Principal Display Panel - 250 mL Bottle Label
-
INGREDIENTS AND APPEARANCE
GLEPTOFORTE
gleptoferron injectionProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:13744-525 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLEPTOFERRON (UNII: 898723IQHQ) (GLEPTOFERRON - UNII:898723IQHQ) GLEPTOFERRON 200 mg in 1 mL Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13744-525-01 100 mL in 1 VIAL, MULTI-DOSE 2 NDC:13744-525-02 250 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200587 09/13/2016 Labeler - Ceva Sante Animale (261126049) Registrant - Ceva Sante Animale (261126049)




