MEDICATION GUIDE
Dispense with Medication Guide available at: www.tevausa.com/medguides
|
Medication Guide ACTIQ® (AK-tik) (fentanyl citrate) oral transmucosal lozenge, CII IMPORTANT: Do not use ACTIQ unless you are regularly using another opioid pain medicine around-the-clock for at least one week or longer for your cancer pain and your body is used to these medicines (this means that you are opioid tolerant). You can ask your healthcare provider if you are opioid tolerant. Keep ACTIQ in a safe place away from children. Get emergency medical help right away if:
These are medical emergencies that can cause death. If possible, remove ACTIQ from the mouth. |
ACTIQ is:
- A strong prescription pain medicine that contains an opioid (narcotic) that is used to manage breakthrough pain in adults (16 years of age and older) with cancer who are already routinely taking other opioid pain medicines around-the-clock for cancer pain. ACTIQ is started only after you have been taking other opioid pain medicines and your body has become used to them (you are opioid tolerant). Do not use ACTIQ if you are not opioid tolerant.
- An opioid pain medicine that can put you at risk for overdose and death. Even if you take your dose correctly as prescribed you are at risk for opioid addiction, abuse, and misuse that can lead to death.
Important information about ACTIQ:
- Get emergency help or call 911 right away if you take too much ACTIQ (overdose). When you first start taking ACTIQ, when your dose is changed, or if you take too much (overdose), serious or life-threatening breathing problems that can lead to death may occur. Talk to your healthcare provider about naloxone, a medicine for the emergency treatment of an opioid overdose.
- Taking ACTIQ with other medicines that may make you sleepy, such as other pain medicines, anti-depressants, sleeping pills, anti-anxiety medicines, antihistamines, or tranquilizers, or with alcohol or street drugs can cause severe drowsiness, confusion, breathing problems, coma, and death.
- Never give anyone else your ACTIQ. They could die from taking it. Selling or giving away ACTIQ is against the law.
- Store ACTIQ securely, out of sight and reach of children, and in a location not accessible by others, including visitors to the home.
- If you stop taking your around-the-clock opioid pain medicine for your cancer pain, you must stop using ACTIQ. You may no longer be opioid tolerant. Talk to your healthcare provider about how to treat your pain.
- ACTIQ is available only through a program called the Transmucosal Immediate Release Fentanyl (TIRF) Risk Evaluation and Mitigation Strategy (REMS). To receive ACTIQ, you must:
- talk to your healthcare provider
- understand the benefits and risks of ACTIQ
- agree to all of the instructions
- sign the Patient Enrollment Form
- ACTIQ is only available at pharmacies that are part of the TIRF REMS. Your healthcare provider can help you locate a pharmacy closest to your home where you can have your ACTIQ prescription filled.
- Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
Do not take ACTIQ if:
- You are not opioid tolerant. Opioid tolerant means that you are already taking other opioid pain medicines around-the-clock for at least one week or longer for your cancer pain, and your body is used to these medicines.
- You have severe asthma, trouble breathing, or other lung problems.
- You have a bowel blockage or have narrowing of the stomach or intestines.
- You are allergic to any of the ingredients in ACTIQ. See the end of this Medication Guide for a complete list of ingredients in ACTIQ.
- You have short-term pain that you would expect to go away in a few days, such as:
- pain after surgery
- headache or migraine
- dental pain
Before taking ACTIQ, tell your healthcare provider if you have a history of:
- troubled breathing or lung problems such as asthma, wheezing, or shortness of breath
- head injury, seizures
- slow heart rate or other heart problems
- low blood pressure
- abuse of street or prescription drugs, alcohol addiction, opioid overdose, or mental health problems
- diabetes. Each ACTIQ unit contains about 1/2 teaspoon (2 grams) of sugar.
- mental problems [including major depression, schizophrenia or hallucinations (seeing or hearing things that are not there)]
- problems urinating
- liver, kidney, thyroid problems
- pancreas or gallbladder problems
Tell your healthcare provider if you are:
- noticing your pain getting worse. If your pain gets worse after you take ACTIQ, do not take more ACTIQ without first talking to your healthcare provider. Talk to your healthcare provider if the pain you have increases, if your feel more sensitive to pain, or if you have new pain after taking ACTIQ.
- pregnant or planning to become pregnant. Use of ACTIQ for an extended period of time during pregnancy can cause withdrawal symptoms in your newborn baby that could be life-threatening if not recognized and treated.
- breastfeeding. ACTIQ passes into breast milk and may harm your baby. Carefully observe infants for increased sleepiness (more than usual), breathing difficulties, or limpness. Seek immediate medical care if you notice these signs.
- living in a household where there are small children or someone who has abused street or prescription drugs.
- taking prescription over-the-counter medicines, vitamins, or herbal supplements. Taking ACTIQ with certain other medicines can cause serious side effects that could lead to death.
When taking ACTIQ:
- Do not change your dose. Take ACTIQ exactly as prescribed by your healthcare provider.
- Your healthcare provider will change the dose until you and your healthcare provider find the right dose for you.
- See the detailed Patient Instructions for Use at the end of this Medication Guide for information about how to use ACTIQ.
- Finish the unit completely in 15 minutes to get the most relief. If you finish ACTIQ too quickly, you will swallow more of the medicine and get less relief.
- Do not bite or chew. You will get less relief for your breakthrough cancer pain.
- You may drink some water before using ACTIQ but you should not drink or eat anything while using ACTIQ.
- You must not use more than 2 units of ACTIQ during each episode of breakthrough cancer pain:
- Use 1 unit for an episode of breakthrough cancer pain. Finish the unit over 15 minutes.
- If your breakthrough cancer pain is not relieved 15 minutes after you finished the ACTIQ unit, use only 1 more unit of ACTIQ at this time.
- If your breakthrough pain does not get better after the second unit of ACTIQ, call your healthcare provider for instructions. Do not use another unit of ACTIQ at this time.
- Wait at least 4 hours before treating a new episode of breakthrough cancer pain with ACTIQ.
- It is important for you to keep taking your around-the-clock opioid pain medicine.
- Talk to your healthcare provider if your dose of ACTIQ does not relieve your breakthrough cancer pain. Your healthcare provider will decide if your dose of ACTIQ needs to be changed.
- Talk to your healthcare provider if you have more than 4 episodes of breakthrough cancer pain per day. The dose of your around-the-clock opioid pain medicine may need to be adjusted.
- If you begin to feel dizzy, sick to your stomach, or very sleepy before ACTIQ is completely dissolved, remove ACTIQ from your mouth.
- Do not stop taking ACTIQ without talking to your healthcare provider. You could become sick with uncomfortable withdrawal symptoms because your body has become used to these medicines. Physical dependency is not the same as drug addiction.
- After you stop taking, or when ACTIQ is no longer needed, see “How should I dispose of ACTIQ units when they are no longer needed?” for proper disposal of ACTIQ.
- Dispose of expired, unwanted, or unused ACTIQ by following the "How should I dispose of ACTIQ units when they are no longer needed?" sections of this Medication Guide below. Visit www.fda.gov/drugdisposal for additional information on disposal of unused medicines.
- DO NOT Drive or operate heavy machinery until you know how ACTIQ affects you. ACTIQ can make you sleepy, dizzy, or lightheaded.
- DO NOT Drink alcohol or use prescription or over-the-counter medicines that contain alcohol. Using products containing alcohol during treatment with ACTIQ may cause you to overdose and die.
- DO NOT Switch from ACTIQ to other medicines that contain fentanyl without talking to your healthcare provider. The amount of fentanyl in a dose of ACTIQ is not the same as the amount of fentanyl in other medicines that contain fentanyl. Your healthcare provider will prescribe a starting dose of ACTIQ that may be different than other fentanyl containing medicines you may have been taking.
The possible side effects of ACTIQ:
- constipation, nausea, sleepiness, vomiting, tiredness, headache, dizziness, abdominal pain, weakness, anxiety, depression, rash, trouble sleeping. Call your healthcare provider if you have any of these symptoms and they are severe.
- Decreased blood pressure. This can make you feel dizzy or lightheaded if you get up too fast from sitting or lying down.
- ACTIQ contains sugar. Cavities and tooth decay can happen in people taking ACTIQ. When taking ACTIQ, you should talk to your dentist about proper care of your teeth.
Get emergency medical help or call 911 right away if you have:
- trouble breathing, shortness of breath, fast heartbeat, chest pain, swelling of your face, tongue, or throat, extreme drowsiness, light-headedness when changing positions, feeling faint, agitation, high body temperature, trouble walking, stiff muscles, or mental changes such as confusion.
- These symptoms can be a sign that you have used too much ACTIQ or the dose is too high for you. These symptoms may lead to serious problems or death if not treated right away. If you have any of these symptoms, do not use any more ACTIQ until you have talked to your healthcare provider.
These are not all the possible side effects of ACTIQ. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. For more information go to dailymed.nlm.nih.gov
How should I store ACTIQ?
-
Always keep ACTIQ in a safe place away from children and from anyone for whom it has not been prescribed. Protect ACTIQ from theft.
- You can use the ACTIQ Child Safety Kit to help you store ACTIQ and your other medicines out of the reach of children. It is very important that you use the items in the ACTIQ Child Safety Kit to help protect the children in your home or visiting your home.
- If you were not offered a Child Safety Kit when you received your medicine, call 888-534-3119.
The ACTIQ Child Safety Kit contains important information on the safe storage and handling of ACTIQ.
The Child Safety Kit includes:
- A child-resistant lock that you use to secure the storage space where you keep ACTIQ (See Figure 1).
Figure 1
-
A portable locking pouch for you to keep a small supply of ACTIQ nearby. The rest of your ACTIQ must be kept in a locked storage space.
- Keep this pouch secured with its lock and keep it out of the reach and sight of children (See Figure 2).
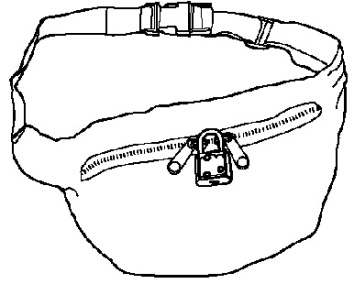
Figure 2
- A child-resistant temporary storage bottle (See Figure 3).
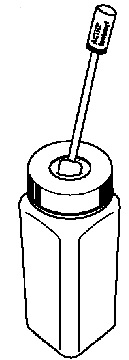
Figure 3
- Store ACTIQ at room temperature, 59°F to 86°F (15°C to 30°C) until ready to use.
- Do not freeze ACTIQ.
- Keep ACTIQ in the original sealed child-resistant blister package. Do not open the blister package until you are ready to use ACTIQ.
- Keep ACTIQ dry.
How should I dispose of ACTIQ units when they are no longer needed?
Disposing of ACTIQ units after use:
Partially used ACTIQ units may contain enough medicine to be harmful or fatal to a child or other adults who have not been prescribed ACTIQ. You must properly dispose of the ACTIQ handle right away after use even if there is little or no medicine left on it.
After you have finished the ACTIQ unit and the medicine is totally gone, throw the handle away in a place that is out of the reach of children.
If any medicine remains on the used ACTIQ unit after you have finished:
- Place the used ACTIQ unit under hot running water until the medicine is gone, and then throw the handle away out of the reach of children and pets (See Figure 4).
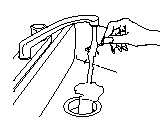
Figure 4
Temporary Storage of Used ACTIQ Units:
- If you did not finish the entire ACTIQ unit and you cannot dissolve the medicine under hot running water right away, put the used ACTIQ unit in the temporary storage bottle that you received in the ACTIQ Child Safety Kit. Push the used ACTIQ unit into the opening on the top until it falls completely into the bottle. Never leave unused or partially used ACTIQ units where children or pets can get to them (See Figure 5).
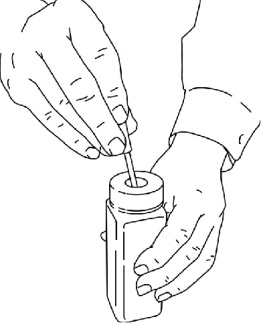
Figure 5
Disposing of Used ACTIQ Units from the Temporary Storage Bottle:
You must dispose of all used ACTIQ units in the temporary storage bottle at least one time each day, as follows:
- To open the temporary storage bottle, push down on the cap until you are able to twist the cap to the left to remove it (See Figure 6).
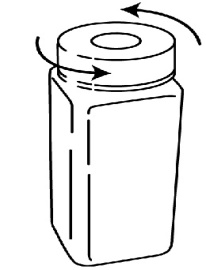
Figure 6
2. Remove one ACTIQ unit from the temporary storage bottle. Hold the ACTIQ by its handle over the toilet bowl.
3. Using wire-cutting pliers, cut the medicine end off so that it falls into the toilet.
4. Throw the handle away in a place that is out of the reach of children.
5. Repeat these 3 steps for each ACTIQ handle that is in the storage bottle. There should not be more than 4 handles in the temporary storage bottle for 1 day.
6. Flush the toilet twice.
Do not flush entire unused ACTIQ units, ACTIQ handles, or blister packages down the toilet.
Disposing of unopened ACTIQ units: Dispose of any unopened ACTIQ units remaining from a prescription as soon as they are no longer needed, as follows:
- Remove all ACTIQ from the locked storage space (See Figure 7).
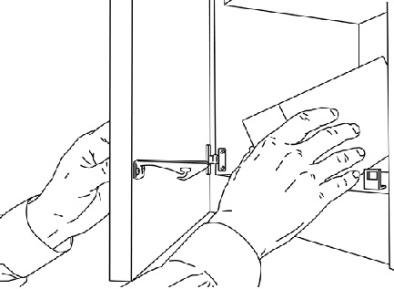
Figure 7
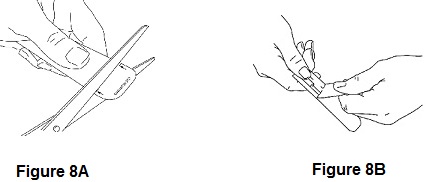
2. Remove one ACTIQ unit from its blister package by using scissors to cut off the marked end and then peel back the blister backing (See Figures 8A and 8B).
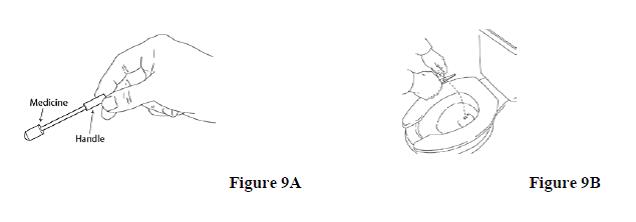
3. Hold ACTIQ by its handle over the toilet bowl. Use wire-cutting pliers to cut the medicine end off so that it falls into the toilet (See Figures 9A and 9B).
4. Throw the handle away in a place that is out of the reach of children (See Figure 10).
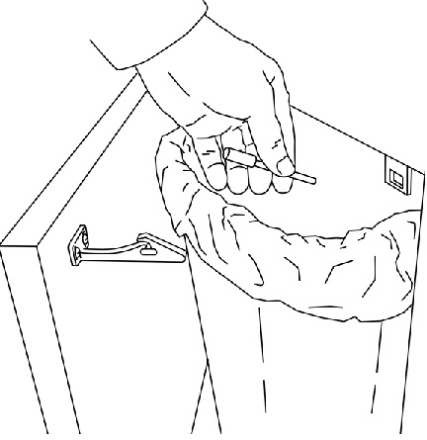
Figure 10
5. Repeat steps 1 through 4 for each ACTIQ unit.
6. Flush the toilet twice after the medicine ends from 5 ACTIQ units have been cut off (See Figure 11). Do not flush more than 5 ACTIQ units at a time.
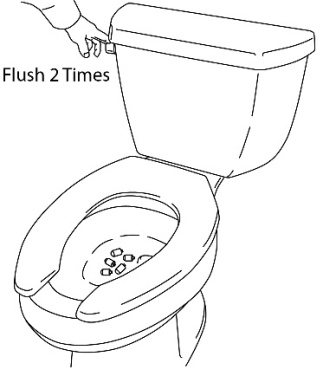
Figure 11
- Do not flush entire unused ACTIQ units, ACTIQ handles, or blister packages down the toilet.
If you need help with disposal of ACTIQ, call Teva Pharmaceuticals at 1-888-483-8279, or call your local Drug Enforcement Agency (DEA) office.
General information about ACTIQ
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Use ACTIQ only for the purpose for which it was prescribed. Do not give ACTIQ to other people, even if they have the same symptoms you have. ACTIQ can harm other people and even cause death. Sharing ACTIQ is against the law.
This Medication Guide summarizes the most important information about ACTIQ. If you would like more information, talk with your healthcare provider or pharmacist. You can ask your pharmacist or healthcare provider for information about ACTIQ that is written for healthcare professionals.
For more information about the TIRF REMS Access program, go to www.TIRFREMSAccess.com or call 1-866-822-1483.
What are the ingredients of ACTIQ?
Active Ingredient: fentanyl citrate
Inactive Ingredients: sugar, citric acid, dibasic sodium phosphate, artificial berry flavor, magnesium stearate, modified food starch and confectioner’s sugar.
Patient Instructions for Use
Before you use ACTIQ, it is important that you read the Medication Guide and these Patient Instructions for Use. Be sure that you read, understand, and follow these Patient Instructions for Use so that you use ACTIQ the right way. Ask your healthcare provider or pharmacist if you have any questions about the right way to use ACTIQ.
When you get an episode of breakthrough cancer pain, use the dose of ACTIQ prescribed by your healthcare provider as follows:
- You may drink some water before using ACTIQ but you should not drink or eat anything while using ACTIQ.
- Each unit of ACTIQ is sealed in its own blister package (See Figure 12). Do not open the blister package until you are ready to use ACTIQ.
Figure 12
- When you are ready to use ACTIQ, cut open the package using scissors. Peel back the blister backing, and remove the ACTIQ unit (See Figures 13A and 13B). The end of the unit printed with “ACTIQ” and the strength number of the unit (“200”, “400”, “600”, “800”, “1200”, or “1600”) is the medicine end that is to be placed in your mouth. Hold the ACTIQ unit by the handle (See Figure 14).
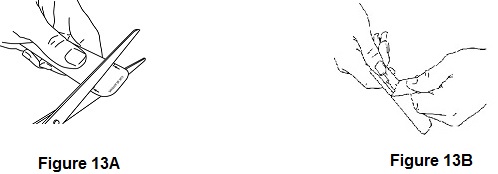
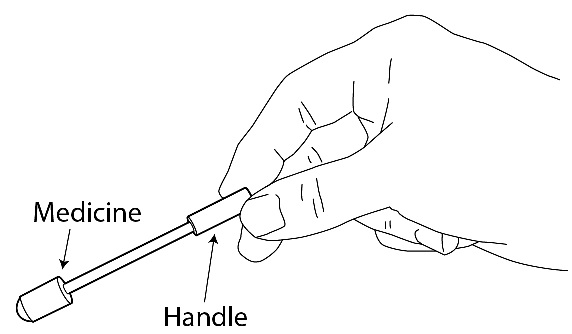
Figure 14
1. Place the medicine end of the ACTIQ unit in your mouth between your cheeks and gums and actively suck on the medicine.
2. Move the medicine end of the ACTIQ unit around in your mouth, especially along the inside of your cheeks (See Figure 15).
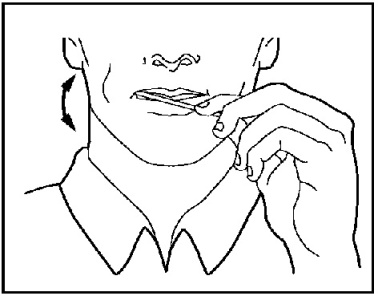
Figure 15
3. Twirl the handle often.
4. Finish the ACTIQ unit completely over 15 minutes to get the most relief. If you finish ACTIQ too quickly, you will swallow more of the medicine and get less relief.
5. Do not bite or chew ACTIQ. You will get less relief for your breakthrough cancer pain.
- If you cannot finish all of the medicine on the ACTIQ unit and cannot dissolve the medicine under hot tap water right away, immediately put the ACTIQ unit in the temporary storage bottle for safe keeping (See Figure 16).
- Push the ACTIQ unit into the opening on the top until it falls completely into the bottle. You must properly dispose of the ACTIQ unit as soon as you can.
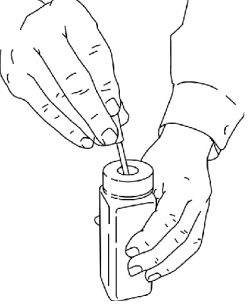
Figure 16
See “How should I dispose of ACTIQ units when they are no longer needed?” for proper disposal of ACTIQ.
|
Distributed by: ACTMG-016 |
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 12/2023 |